Chief Complaint: Worsening vision and progressive corneal ectasia in a patient with keratoconus.
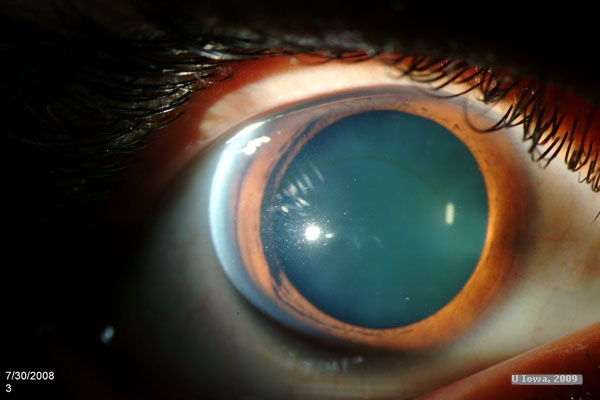
History of Present Illness: A 22-year-old male with keratoconus was referred for progressive corneal ectasia and worsening visual acuity. Initial management included spectacle correction, but progression of his disease required correction with rigid gas permeable contact lenses. Best corrected visual acuity was 20/100 OD, 20/50 OS and 20/50 OU. The patient was unable to work because of poor vision. Glasses did not provide adequate correction and he was intolerant of contact lenses, particularly in the right eye in which the lens continued to pop out despite the best possible fit. The patient was referred to the University of Iowa Cornea Service for consideration of surgical intervention.
Past Ocular History: Progressive keratoconus OD > OS as described above.
Medical History: None.
Family History: Father with keratoconus.
Medications: none
 |
 |
The most common surgical options for keratoconus include Deep Anterior Lamellar Keratoplasty (DALK) and Penetrating Keratoplasty (PK):
-Penetrating keratoplasty (PK)
Penetrating keratoplasty is a well-studied and long established surgery for the treatment of corneal disease such as keratoconus. However, replacement of donor endothelium makes host rejection of the graft more likely. A 15-year study of the outcomes of PK demonstrated graft failure in 76 out of 500 eyes, with rejection accounting for 25% of those failures. The overall probability of developing graft rejection was 23%. Late endothelial failure caused 29% of graft failure, (4) suggesting that DALK, which retains native endothelium, may increase long term graft survival.
-Deep anterior lamellar keratoplasty (DALK)Deep anterior lamellar keratoplasty has been proposed as an excellent alternative to penetrating keratoplasty for corneal diseases that do not affect the endothelium. DALK preserves native endothelium and reduces host immune system reaction and graft rejection. In a recent study of outcomes using the "big bubble" technique, only 2 of 78 patients developed stromal rejection. (1) This fits well with other studies reporting rejection in 3-8% of cases (2).
In addition, a 2 year retrospective study by Rice found significantly less astigmatism in patients undergoing DALK as opposed to PK. However, the same study reported slightly better visual outcomes at 2 and 4 year in the PK group. Average best-corrected visual acuity 4 years post-graft was 6/6 in the PK group and 6/7.5 in the DALK group. (3)
In a study comparing DLK with PKP, both procedures resulted in good visual and refractive outcomes. 95% of PKP patients achieved 6/12 vision or better postoperatively. 88% of patients with DLK achieved vision of 6/12 or better by 8 months. Median spherical equivalent for DALK was -4.13 diopters (-0.50 to -8.38). Median spherical equivalent for PKP was -1.63 diopters (-16.00 to +4.00). Complication rates were similar between the two surgeries, however, this was noted to be dependent on the skill of the surgeon. The encountered complications were less serious in the DALK group. (2)
| Penetrating keratoplasty | Deep anterior lamellar keratoplasty |
|---|---|
Pros:
Cons:
|
Pros:
Cons:
|
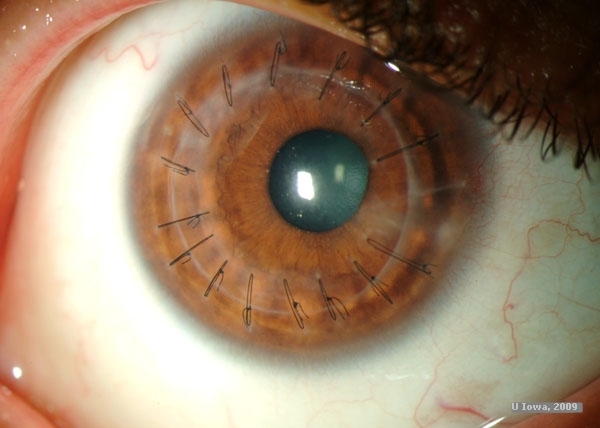
DALK using the "Big Bubble" technique as described by Anwar was recommended and successfully performed on our patient’s right eye. Two months post surgery, uncorrected acuity was 20/250. At 4 months post surgery, uncorrected visual acuity improved to 20/100, with best corrected vision of 20/60. At 6 months post surgery, the patient was fitted for contact lenses and ultimately achieved visual acuity of 20/50. Of note, OS vision has progressively declined, ultimately to a best corrected acuity of 20/80. Consequently, DALK has been scheduled for the left eye.
 |
Keratoconus (KCN) is a progressive, non-inflammatory ectatic corneal disorder. It is commonly bilateral. The disease progresses throughout the second and third decades of life. Its etiology is unknown.
Degradative enzymes in the cornea have been shown to be elevated in KCN with a concurrent down regulation of proteinase inhibitors. Microtrauma, possibly from eye-rubbing and contact lens wear, has been suggested as a contributor to keratoconus. Eye rubbing has been reported in 66-73% of patients with KCN. A history of contact lens wear has been reported in 17-26% or patients with KCN. Keratoconus is associated with collagen disorders such as Ehlers-Danlos and Osteogenesis Imperfecta. KCN is also has a higher incidence in patients with Down’s Syndrome.(5)
Discordance between dizygotic and monozygotic twin studies suggest a genetic role in the development of KCN. A family history of KCN is common, as was the case with our patient and his affected father. The disease is multifactorial in origin. Many gene loci have been implicated in the development of KCN. These include loci in the chromosomal regions 2p24, (6) 3p14-q13, (7) 5q14.3-q21.1 (8), and 16q22.3-23.1 (9), identified in large single or multiple pedigrees by genome wide-mapping and suggesting an autosomal dominant model of transmission. Also identified by affected-only linkage analysis, indicating multifactorial transmission, are the chromosome regions 4q31, 5q31, 9q34, 12p12, 14p11, 17q24 and 20q12.(10,11) The visual system homeobox gene VSX1 on chromosome 20 has also been indicated in studies.(12, 13) Despite its higher incidence in patients with Trisomy 21, no loci on this chromosome have been identified.
All layers of the cornea appear to be affected. Degeneration of basal epithelial cells allows anterior collagen growth into the epithelium and posterior epithelial growth into Bowman’s layer, forming "Z" shaped lesions typical of keratoconus. Ferritin deposition in the basal epithelial cells (Fleischer ring) is a hallmark of the disease, and as ectasia progresses this ring becomes dense and narrow. Areas of clearing in the stroma are typical. Keratocyte density is decreased. Thinning of the stroma allows the cornea to take on a more conical shape, usually with the apex of this cone just inferior to the central visual axis. As a result, patients experience mild to severe impairment of visual acuity from irregular astigmatism.(5)
The diagnosis is made clinically. Patients will have high, irregular astigmatism with scissoring seen on retinoscopy. Keratometry shows corneal steepening, commonly inferior. Slit lamp examination shows protrusion of the cornea with thinning of the apex, typically from ½ to 1/5th the normal thickness of the cornea. Striae are seen anterior to Descemet’s membrane (Vogt’s striae), and may resolve with external compression near the limbus. Superficial scarring of Bowman’s layer is often present. The red reflex may be abnormal. In some cases, corneal hydrops may occur, caused by disruptions in Descemet’s membrane and causing sudden vision loss.(5)
EPIDEMIOLOGY
|
SIGNS
|
SYMPTOMS
|
TREATMENT
|
Raecker M, Goins K: Verticillata: Surgical Management of Keratoconus: A 22-year-old male with a history of progressive keratoconus. EyeRounds.org. October 5, 2009; Available from: http://www.EyeRounds.org/cases/98-Keratoconus-Surgical-Options.htm.

Ophthalmic Atlas Images by EyeRounds.org, The University of Iowa are licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License.