


INITIAL PRESENTATION
Chief Complaint: Blurry vision in the left eye and headaches.
History of Present Illness:
A 44-year-old female presents with a four-week history of headaches, vision changes, and tinnitus. She reports her headaches vary in location but typically begin unilaterally and progress to bilateral involvement with accompanying visual aura including zig zags, photophobia, phonophobia, and nausea. Although she has experienced these headaches every few days over the past seven years, they have now been occurring daily for the past few months. A week ago, she also noticed that her left eye had a large black spot in the middle of her visual field obscuring about 50% of her vision. She currently endorses some waxing and waning of the size and shape of the black spot in her vision. She is also experiencing some rainbow bursts and notes many small floaters in both eyes, though she describes the floaters more like TV static. Her peripheral vision is otherwise subjectively intact. She reports a previous episode of spontaneous bilateral vision loss lasting 2 minutes in 2018. Regarding the patient’s auditory symptoms, she reports a constant “swooshing” sound in her left ear for the past 3 months. She also had a previous episode of hearing loss in her right ear in 2018 which resolved spontaneously after 9 months. She has no focal weakness, slurred speech, or confusion.
Past Ocular History:
Past Medical History:
Medications:
Allergies:
Family History:
Social History:
Review of Systems:
OCULAR EXAMINATION
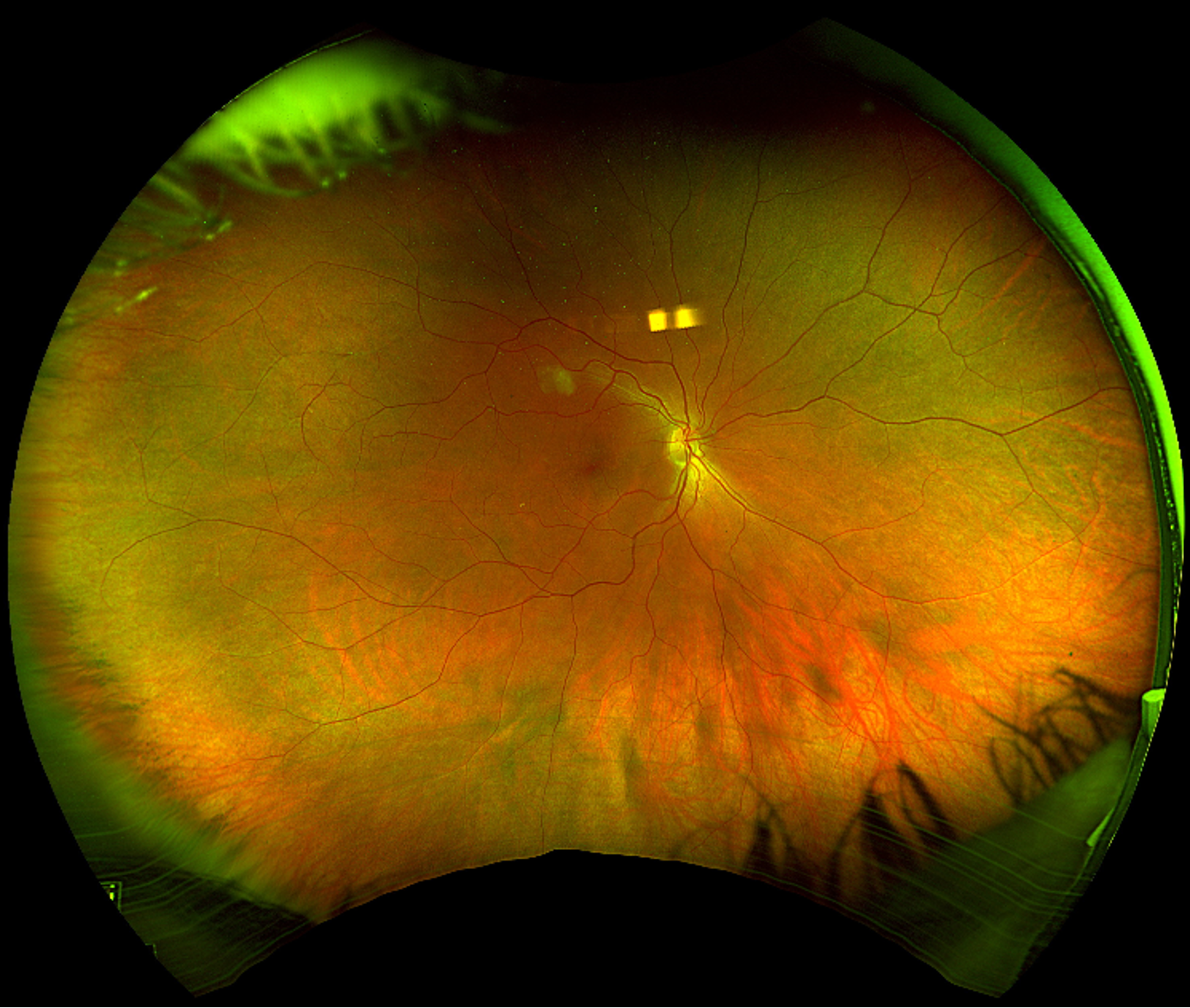
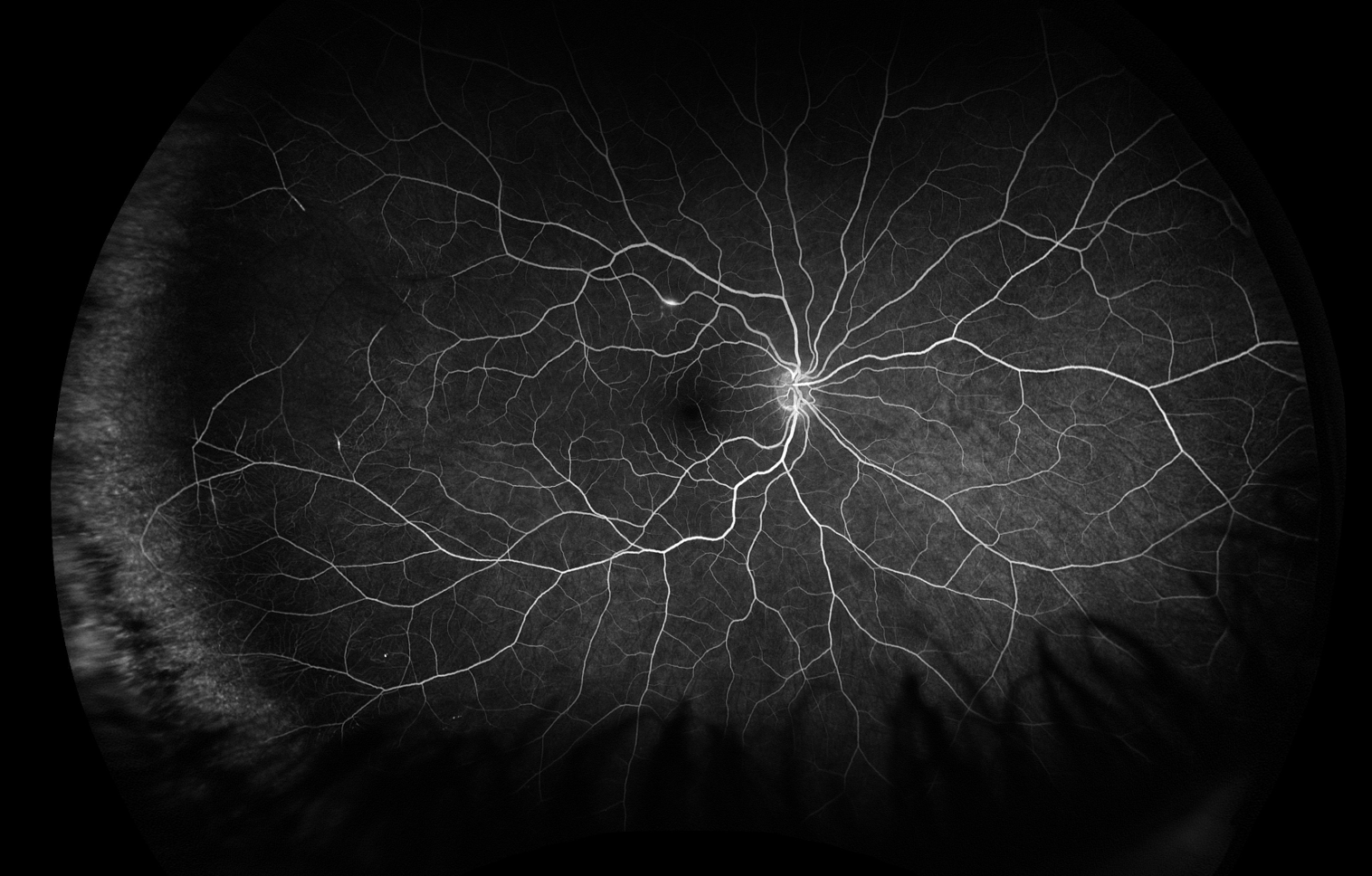
DIFFERENTIAL DIAGNOSIS:
CLINICAL COURSE
The patient was initially treated for possible stroke vs MS flare at the outside hospital where she was started on aspirin, Plavix, and atorvastatin and IV steroids. Brain MRI with contrast was performed and revealed both acute and chronic lesions in the corpus collosum and internal capsule. Further imaging done at the outside hospital included MRA head/neck without contrast, TTE, TEE, and venous duplex of all four extremities. TTE revealed a patent foramen ovale. The patient was still having left eye vision changes, severe headaches, and episodes of whole-body weakness, so she was transferred to UIHC.
At UIHC, the patient was admitted to the Neurology service and was started on a 7-day course of 1g daily IV methylprednisone. Ophthalmology was consulted, and initial exam showed a single branch retinal artery occlusion along the superior arcade of the right eye and multiple branch retinal artery occlusions in the left eye. She was diagnosed with Susac syndrome and she continued to follow with neurology for a 6-week prednisone taper (starting at 60 mg and decreasing by 10 mg each week) and planned initiation of Rituximab injections in 1 month.
Five weeks later, the patient presented again to the UIHC emergency department with worsening symptoms after tapering oral prednisone to 30 mg. She had been unable to start Rituximab injections due to difficulties with insurance approval. At this time, she was admitted to the hospital again and started on IVIG 2 g/kg over 3 days followed by 40 mg of oral prednisone daily. Ophthalmology was again consulted and determined that her visual acuity had improved in the left eye from the previous admission and there were no new BRAOs. She was discharged on 40 mg prednisone daily with a plan for Rituximab injection in 2 weeks pending insurance approval.
She was seen for 2 and 4 month follow up in retina clinic, with recommendation to continue steroid taper and up-titrate Rituximab to maintenance levels. Continued monitoring with repeat fluorescein angiography was then performed at 4-6 month intervals.
DIAGNOSIS: Susac Syndrome
DISCUSSION
Etiology/Epidemiology:
Susac syndrome is a rare autoimmune microangiopathy associated with a classic triad of encephalopathy, branch retinal artery occlusion, and sensorineural hearing loss (1). Due to its rarity, the true incidence of Susac syndrome is unclear, however data from small Austrian and Israeli case series estimate an annual incidence between 0.024 and 0.13 per 100,000 individuals respectively (2,3). There is a female predominance, with a female-to-male ratio of 3.5:1, and it most commonly presents in individuals aged 16 to 40 years, though it can occur across a wider age range (1).
The clinical course of Susac syndrome varies widely, and can be classified as monocyclic, polycyclic, or chronic–continuous. Monocyclic disease is defined by a self-limiting course with an active period lasting ≤2 years, whereas polycyclic disease involves one or more relapses after 2 years from onset. Thus, chronic-continuous disease includes >2 years of active symptoms without remission. Using the above parameters, it is estimated that 54% of patients develop monocyclic disease, 42% polycyclic disease, and 4% chronic–continuous. Of note, the same 2008 review of all published cases worldwide at the time found that only 13% of patients presented with the full triad of symptoms and the average time between symptom onset and completion of the triad was 21 weeks (1).
Pathophysiology:
Susac syndrome is characterized by damage to microvascular endothelial cells leading to thrombotic occlusion of small arterioles in the brain, retina, and inner ear (4).
Although the exact etiology remains unclear, it is believed to involve an autoimmune mechanism potentially linked to anti-endothelial-cell antibodies (AECA). Pathologic studies consistently indicate a T-cell mediated process, targeting the microvasculature of the brain, retina, and inner ear, where antibodies target endothelial cells, leading to inflammation and subsequent occlusion of the vessels. One study found that approximately 30% of patients with Susac syndrome had positive serum titers for AECA (5). Deposition of complement factor C4d within capillary walls has been demonstrated on brain biopsy, supporting an antibody-mediated vascular injury (6). However, anti-endothelial-cell antibodies are not sensitive or specific for Susac syndrome and may not have a pathogenic role in the disease, as it is possible they may develop following endothelial cell damage (7). Recent research suggests CD8+ T-lymphocytes may drive the endotheliopathy as a cytotoxic response to an antigen expressed on endothelial cells (8). Regardless, an autoimmune response with microvascular thrombosis leads to ischemic damage in affected tissues, manifesting as encephalopathy, branch retinal artery occlusions (BRAO), and sensorineural hearing loss.
Signs/Symptoms:
Susac syndrome manifests with a distinctive triad of symptoms: encephalopathy, branch retinal artery occlusions, and sensorineural hearing loss. Though over the course of the disease, 85% of patients develop the complete triad of manifestations, as mentioned, only 13% of patients present with the full triad (1). Encephalopathy in Susac syndrome can manifest as headaches, confusion, memory loss, and psychiatric disturbances, reflecting the involvement of the brain’s microvasculature. Headache is the most common reported symptom and is seen in 80% of patients (1). Symptoms from branch retinal artery occlusions typically range from no symptoms to visual disturbances such as scotomas, blurring, or even partial vision loss attributable to impaired blood flow within the retinal arteries. Sensorineural hearing loss often occurs suddenly and can affect one or both ears, leading to varying degrees of hearing impairment (1).
Testing/Imaging/Laboratory work-up:
Magnetic resonance imaging is the imaging modality of choice for Susac syndrome; however, its findings can be difficult to distinguish from other conditions that can cause white matter lesions such as Multiple Sclerosis (MS) and Acute Disseminated Encephalomyelitis (ADEM). In addition to similar imaging findings, these conditions can have overlapping clinical presentations and are more common in comparison, sometimes leading to misdiagnosis of Susac syndrome. On MRI, Susac syndrome typically reveals multifocal lesions in the central corpus callosum, appearing as distinct, round "snowballs" on T2-weighted and FLAIR imaging. Punctate microinfarcts within the internal capsule forming a characteristic “sting of pearl” appearance are also characteristic of Susac syndrome (9).
These round, snowball lesions on MRI are pathognomonic for Susac syndrome. In contrast, typical MS lesions known as Dawson’s fingers are typically ovoid and are limited to white matter involvement. Susac syndrome, unlike MS and ADEM, can feature both parenchymal and meningeal enhancement, and its white matter lesions lack the central vein often seen in MS.
Additionally, the central placement of the lesions in the corpus callosum can aid in distinguishing MRI findings, as lesions in ADEM and MS are typically location along the under-surface and septal interface of the corpus callosum (10).
Fluorescein angiography in Susac syndrome often reveals multiple branch retinal artery occlusions (BRAO), which can also be visualized on fundoscopic examination and optical coherence tomography. In addition to BRAO, characteristic ophthalmologic findings in Susac syndrome include Gass plaques, visible on fundoscopic exam, and arteriolar wall hyperfluorescence (AWH), visible on fluorescein angiography (11). Gass plaques are yellow refractile or non-refractile lesions along the arterioles attributed to lipid deposition occurring in regions of damaged endothelial cells, and their appearance may fluctuate throughout the disease course. Retinal emboli, whether platelet-fibrin, calcific, or cholesterol (Hollenhorst) plaques, frequently lodge at branch points in the arterial vasculature. Conversely, Gass plaques are often located along arteriole straight segments (12). AWH results from fluorescein staining of damaged vessel walls and is a hallmark of Susac syndrome, especially when observed in normal-appearing vessels distant from areas of BRAO. AWH can persist even during inactive disease and, when located away from branch points, is nearly pathognomonic for Susac syndrome (11).
Additional testing may include audiometric testing and cerebrospinal fluid (CSF) analysis. Audiometric testing frequently identifies sensorineural hearing loss, reflecting cochlear microangiopathy. CSF analysis can be used to rule out other differential diagnoses but is not diagnostic for Susac syndrome (9).
Treatment/Management/Guidelines:
There is no one standardized treatment of Susac syndrome primarily due to its relative rarity preventing the performance of any randomized control trials to date. However, treatment generally requires a regimen of immunosuppressant medications, tailored to the severity and distribution of the disease - central nervous system, retina, and/or inner ear (13).
The most recent guidelines for treatment of Susac syndrome recommend stratifying the treatment approach based on the severity of CNS involvement – ranging from mild to extremely severe cases. Corticosteroids (IV methylprednisolone 1000 mg/day for 3-7 days, followed by oral prednisone 1 mg/kg/day for 4 weeks then tapered) form the basis for treatment across all severity levels. IVIG is used additionally, typically starting with 2 gm/kg over 2 days, followed by a lower maintenance dose, depending on the severity and response to treatment (12a1). Cyclophosphamide is included in the treatment regimen for extremely severe and severe cases but is optional for moderate cases and not typically recommended for mild cases. For long-term immunosuppression, mycophenolate mofetil is a standard option and is often used alongside tacrolimus in more severe cases. Rituximab may also be used, especially in more severe presentations, with repeat doses as needed based on disease control. The duration of treatment varies, but maintenance immunosuppression is typically required for at least two years with frequent monitoring and adjustments based on disease progression (13).
While retinal vasculopathy and inner ear disease often present concurrently with CNS Susac syndrome, they may also manifest as the primary symptom. When these conditions accompany CNS Susac syndrome, treatment generally aligns with the guidelines discussed above. However, if they occur independently, treatment may not need to be as aggressive or for as long of a duration (13).
Each patient’s treatment plan will vary based on the specific presentation and response to treatment, emphasizing the importance of close monitoring with serial examination to help guide appropriate medication adjustments. In addition to ophthalmologic examination, monitoring typically involves MRIs, fluorescein angiography studies, and audiograms, along with thorough documentation of the patient’s symptoms.
Early diagnosis and immunosuppressive treatment are crucial to mitigate the progression of the disease and prevent long-term complications such as blindness, deafness, and cognitive impairment. Overall, patients with Susac Syndrome generally have a favorable long-term prognosis.
EPIDEMIOLOGY OR ETIOLOGY
|
SIGNS
|
SYMPTOMS
|
TREATMENT/MANAGEMENT
|
Jones G, Meyer M, Hunt M, Russell J. Susac Syndrome. EyeRounds.org. February 24, 2026. Available from https://EyeRounds.org/cases/376-susac-syndrome.htm

Ophthalmic Atlas Images by EyeRounds.org, The University of Iowa are licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License.