Chief Complaint: 22-year-old Caucasian female referred in 1990 for evaluation of elevated intraocular pressure (IOP) that was initially controlled with medicines.
History of Present Illness: High IOP was incidentally detected in both eyes at age 16 and was effectively controlled until 6 months ago, when IOPs reached the mid-50's OU.
Ocular History: No previous ocular trauma or surgery.
Medical History: Unremarkable.
Medications:
Social History: Noncontributory.
Family History: mother, grandmother, and brother have glaucoma.
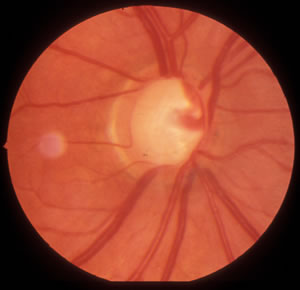 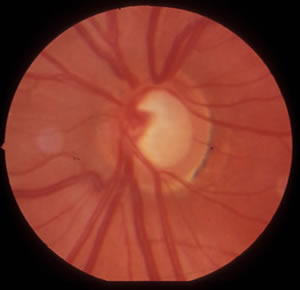 |
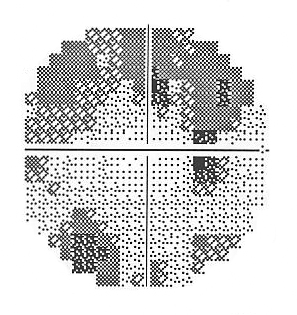
| A. The visual field test result from the right eye shows a superior arcuate scotoma, and the beginnings of an inferior arcuate scotoma. | B. The visual field test result from the left eye also shows superior and inferior arcuate scotomas that are consistent with glaucoma. |
 |
 |
 |
Juvenile open-angle glaucoma (JOAG) is a rare form of glaucoma that differs from adult-onset primary open angle glaucoma (POAG) in its age of onset and often in the magnitude of IOP elevation. By definition JOAG has its onset between 3 and 40 years of age, while POAG has its onset after the age of 40. Also, patients with JOAG often have extremely high IOP, sometimes greater than 50 mm Hg. The genetic basis of JOAG is much more obvious than that of POAG. JOAG is frequently passed down through families as an autosomal trait (Fingert, 2002).
Many cases of JOAG are caused by mutations in the myocilin gene, especially subjects that have early-onset of disease, high IOP, and a strong family history of glaucoma. As many as 8-63% of JOAG cases are associated with myocilin mutations (Wiggs, 1998, Adam, 1997) while the fraction of cases due to myocilin mutations may be much higher when there is a strong family history (Stone, 1997, Alward, 1998).
JOAG is treated using the same medical and surgical therapies as POAG, however, medical therapies often have had limited success in managing JOAG. Many JOAG patients require surgical therapy for adequate control of IOP (Stokes, 1940; Johnson, 1993)
One set of mutations in the myocilin gene (for example PRO370LEU, TYR437ILE, ILE477ASN) is associated with JOAG while others (GLN368STOP) are associated with late-onset POAG. Mutation-specific features of glaucoma such as age of onset and maximum IOP has been described for many myocilin mutations (Alward, 1998; Fingert, 2002).
Because early clinical signs of glaucoma are subtle, the diagnosis may often be delayed until the disease has progressed. Genetic testing for a myocilin mutation has the potential to aid in early diagnosis of glaucoma and may have clinical utility in specific circumstances. There is a reasonably high likelihood that patients with JOAG have disease due to myocilin mutations, especially when there is an early age at onset, marked elevation in IOP, and strong family history. Patients with these features may benefit from genetic testing and should discuss this option with their ophthalmologist and/or genetic counselor. Information about genetic testing for myocilin worldwide is available at GeneTests (www.genetests.org). At present, the John and Marcia Carver Nonprofit Genetic Testing Laboratory at the University of Iowa (www.carverlab.org) is the only laboratory offering diagnostic testing for myocilin mutations in the United States.
A series of medical therapies including the available topical medications in 1990 (Timolol, dipivefrin, pilocarpine) and oral medications (acetazolamide) were unable to control IOP. As medical therapies failed, surgical options were considered early in her course. Trabeculectomies were eventually performed in both eyes with good pressure control.
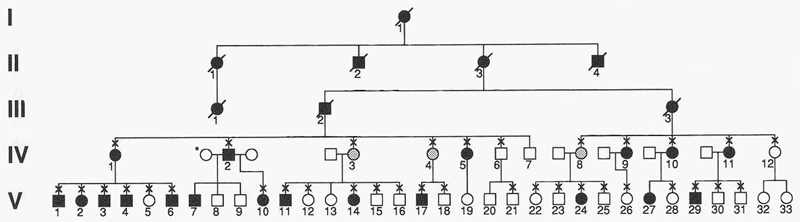
The patient was found to have a strong family history of JOAG with over 25 affected family members (Figure 3). Blood samples were obtained from affected family members and genetic testing revealed a TYR437HIS mutation in the patient as well as all other family members that are affected with JOAG. Genetic testing for this family was provided and used to help identify those family members at greatest risk for developing glaucoma so that appropriate medical and surgical options could be offered without delay. Also, those family members with no myocilin mutations were reassured that they were not at increased risk for developing JOAG, however, they still had the population risk of developing glaucoma that is unrelated to mutations in the myocilin gene.
Diagnosis: Juvenile Opem-Angle Glaucoma (JOAG)
EPIDEMIOLOGY
|
SIGNS
|
SYMPTOMS
|
TREATMENT
|
Lively GD, Alward, WL, Fingert JH. Juvenile open-angle glaucoma: 22-year-old Caucasian female referred in 1990 for evaluation of elevated intraocular pressure (IOP). EyeRounds.org. September 17, 2008; Available from: http://www.EyeRounds.org/cases/87-Myocilin-juvenile-open-angle-glaucoma.htm.

Ophthalmic Atlas Images by EyeRounds.org, The University of Iowa are licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License.