
The University of Iowa
Department of Ophthalmology and Visual Sciences
January 6, 2015
Treatment of retinal disease by intravitreal injection (IVI) has revolutionized the field of ophthalmology. It is estimated that over four million IVIs were performed in the United States in 2013, a number that is expected to continue to increase [1]. This procedure, considered a subspecialty skill, is increasingly performed in the office of comprehensive ophthalmologists, especially for the treatment of common eye diseases like choroidal neovascularization in age-related macular degeneration. Given the risk of complications, especially endophthalmitis and suprachoroidal hemorrhage, and the need to handle these complications, IVI by generalists is controversial and even opposed by some vitreoretinal specialists [2]. While this "scope of practice" debate continues on, ophthalmology residency programs are now training their residents to properly perform IVIs as part of a comprehensive curriculum. Unfortunately, a wide variety of IVI protocols exist in terms of aseptic technique, anesthetics, use of prophylactic antibiotics, and post-injection monitoring.
This article is intended to be a resource and primer for ophthalmology residents learning IVI. We aim to present a helpful, concise review of recent evidence surrounding IVI technique and describe IVI protocols at the Iowa City Veterans Affairs hospital and the University of Iowa.
Common complications of IVI are injection site discomfort, subconjunctival (SC) hemorrhage, vitreous reflux, and transient elevation of intraocular pressure (IOP). Patients may also develop floaters, vitreous or retinal hemorrhage, and retinal detachment. The most dreaded complication of IVI, though rare, is endophthalmitis (EO), and much of IVI protocol is designed to minimize, or eliminate, EO due to human error. EO rates reported in large multi-center randomized controlled trials of anti-vascular endothelial growth factor (anti-VEGF) IVI range from 0.019% [3] to 0.09% [4] per injection.
Close examination of the events surrounding historical large outbreaks of infectious EO suggest that these outbreaks were not a result of the drug or the injection technique, but rather of the pharmacological compounding procedures used to prepare the syringes containing the drug. Noncompliance with standards and poor aseptic technique were the most likely cause, and implementation of and strict adherence to United States Pharmacopoeia requirements has since been emphasized [5]. "Areas for improvement" that remain are much of what this article will discuss.
IVIs are generally performed in the office. In fact, many papers on IVI do not mention the setting, as in-office IVI is assumed. However, performing IVI in the operating room (OR), a more controlled and sterile environment, may decrease EO incidence… but at considerable cost.
EO rates after IVI performed in the OR are very low. In 2014, a retrospective study of two Swiss eye hospitals of 40,011 IVIs, all performed in the OR, reported just three cases of EO for a rate of 0.0075% [6]. The only major procedural difference between the two hospitals was the use of post-operative antibiotics at one hospital, accounting for approximately 13,337 IVIs and two of the three EO cases. This study was limited by its lack of a control group. These EO rates for IVI performed in the OR could be loosely compared to the prospective and randomized CATT 2-year results of 12,886 office-based injections where EO rates were approximately 0.09% [7]. A retrospective series of 14,895 office-based IVIs, by Cheung et al., reported EO rates of 0.057% [8]. Another retrospective study of 10,254 office-based IVIs, by Pilli et al., reported 0.029% [9].
A 2012 report of a single-surgeon cohort study from Australia of 12,249 IVIs showed a statistically significant difference in EO incidence rates for IVIs performed in the office procedure room (0.12%) versus OR (0%) [10]. However, results were limited by a lack of randomization.
A more recent 2014 retrospective review by Tabandeh et al. compared EO rates for all patients who underwent IVI from 2009-2011 by two ophthalmologists (retinal specialists), one who performed IVI in the office and the other in the OR [11]. They included 11,710 IVI: 8,647 office-based and 3,063 performed in the OR. Of note, office-based IVI were done without a mask and with no limitation on conversation. There was no significant difference in the rate of EO between the office-based (0.035%) and OR (0.065%).
Based on the most recent data, OR-based injections have very low rates of EO. These rates, however, do not appear to be significantly less than rates for office-based injections. A large, randomized controlled trial comparing the two settings has yet to be published. Moving IVI to the OR would increase running costs, time per patient treated, and overall inconvenience to the patient. With so many patients requiring IVIs, the need to treat the underlying disease may outweigh the additional risk profile of in-office injections. For now, the majority of IVIs are performed in the office.
The main goal of lid retraction is to avoid needle contamination by the lashes via involuntary/reflex lid closure during needle insertion. The first anti-VEGF study clearly showed an increase in complications when no retractor was used [12]. Sterility, ease of use, exposure, and patient comfort are all considerations. The majority of ophthalmologists use metal lid retractors [13], but several other mechanisms have been proposed, such as bimanual assisted eyelid retraction [14], conjunctival mold [15], upper lid retraction using a Desmarres retractor [16] and most recently, cotton-tipped applicator lid retraction [17]. No literature compares various retraction techniques and post-IVI complications, particularly EO.
The main goal of lid retraction is to avoid needle contamination by the lashes via involuntary/reflex lid closure during needle insertion. The first anti-VEGF study clearly showed an increase in complications when no retractor was used [12]. Sterility, ease of use, exposure, and patient comfort are all considerations. The majority of ophthalmologists use metal lid retractors [13], but several other mechanisms have been proposed, such as bimanual assisted eyelid retraction [14], conjunctival mold [15], upper lid retraction using a Desmarres retractor [16] and most recently, cotton-tipped applicator lid retraction [17]. No literature compares various retraction techniques and post-IVI complications, particularly EO.
The primary goals of anesthesia are 1) prevent patient movement and involuntary lid closure during needle insertion and 2) increase patient comfort and patient compliance for a procedure that needs to be repeated frequently. Many approaches to ocular surface anesthesia have been reported and include topical drops, anesthetic-soaked applicators, gels, and SC injection of anesthetics. Most reported data show: 1) Topical anesthesia and SC anesthesia are no different, but SC anesthesia may have more side effects like SC hemorrhage. 2) Various forms of topical anesthesia are no different.
A study by Blaha et al. demonstrated no difference in pain score between proparacaine drops, tetracaine drops, lidocaine-soaked, cotton-tipped swabs, and SC lidocaine. SC lidocaine had the most side effects [18]. Another study found IVI to be less painful with SC injection, but when the pain from the SC injection was included, pain scores were no different than with topical anesthesia [19]. A study comparing proparacaine 0.5% drops, proparacaine + 4% lidocaine-soaked, cotton-tipped swabs, and 3.5% lidocaine gel found no difference in pain or burning scores [20].
One concern is that gel anesthetic may reduce the efficacy of ocular surface antisepsis (discussed in the next section). When povidone-iodine (PI) and lidocaine gel are inoculated into culture media with bacteria, the same amount of bacteria grow as with lidocaine gel alone [21]. Another similar study confirmed these results but also found that PI applied for 5-30 seconds prior to lidocaine gel is as effective as PI alone [22]. Meaning, if gel is used, it should be applied after PI.
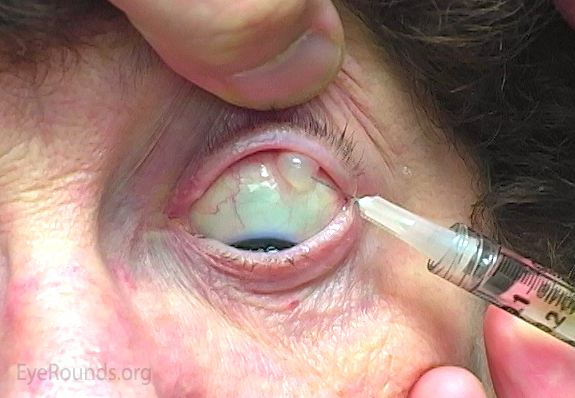
In a 2011 survey of 765 retinal specialists, > 99% use PI prior to IVI [13]. It is cheap, has broad-spectrum coverage over a range of concentrations, a fast kill-time (15-120 seconds), no reported resistance, and no reports of anaphylaxis [23]. If it is inadvertently "pushed" into the eye during injection, it should cause no harm to the eye [24].
While chlorhexidine may be more effective than PI when used in other settings, alcoholic chlorhexidine is toxic to the corneal epithelium. Aqueous chlorhexidine is safe and could be used in the setting of PI allergy [29].
Streptococci are reported to comprise 7% of the conjunctival flora [30]. In a report of 52 cases of EO, occurring in 105,536 IVIs, Staphylococci were responsible for 65% of cases and streptococci 31% [31]. A 2011 meta-analysis of most major US studies from 2005-2010 of EO after IVI of anti-VEGF agents reported streptococcal species were approximately three times more commonly the cause of post-IVI EO than post-intraocular surgery, where masks are worn [31]. This over-representation of streptococcus is believed to come from the oral cavity of either healthcare providers or the patient.
Wen et al. simulated an IVI under different conditions and found significantly more colony-forming bacteria are dispersed onto an agar plate when patients were speaking without a face mask compared with when wearing a face mask or remaining silent. They also found that speaking in a reclined position (in an ophthalmic exam chair, reclined so that the patient's face is parallel to the floor) results in significantly MORE colony-forming bacteria on the culture plate (placed on the forehead) than on the background control plate [32]. This indicates that a fully reclined, unmasked patient may disperse bacteria towards the eyes. There is currently no published data on masked and reclined patients and whether this would increase or decrease contamination. In the OR, a patient's nasopharyngeal area is typically covered by an adhesive drape to isolate the eye and periocular region. It is unlikely that a simple surgical mask would be as efficacious.
A study of ten surgeons reciting a 30-second script in four different scenarios compared bacterial growth on a blood agar plate with no facemask, the use of a standard surgical facemask, no mask but 5% PI pre-treated plate, and no mask but silence. There was significantly less bacterial growth for the facemask group and silence group compared to no mask, but pre-treated PI plates, even without facemask, demonstrated the least bacterial growth overall [33]. Conversely, a study of needle contamination between unmasked talking versus silent breathing over a sterile needle for 30 seconds found no significant difference between needle cultures, suggesting there may be no need for silence during IVI [34]. This study, however, was limited by a very small sample size.
Shimada et al. published an efficacy report for IVI protocol at a single university hospital, where all anti-VEGF IVIs over 3 years (15,144 injections) were done with doctors and nurses wearing surgical masks, eyelid skin disinfected with 10% PI and conjunctiva with 0.25% PI, patients' faces were draped, and the conjunctival surface was washed with 5 ml of 0.25% PI, waiting 30 seconds before IVI. Post-IVI site was washed again and patients received three days of levofloxacin. EO rates were zero. Without a control group, unfortunately, it is impossible to know if results are related to masks, draping, antibiotics, or some other variable [35].
Despite simulation studies demonstrating reduced contamination of culture plates with masking and silence, wearing a facemask is still not considered a uniform standard in the ophthalmology community. Because of the shear volume of IVI annually, the addition of facemasks for physicians and staff assisting in IVI would amount to an astounding increase in healthcare costs, up to $1.5 million annually [35].
A variety of options exist. The needle gauge used for IVI, the angle of needle insertion (or "incision"), the depth of insertion, and the speed of insertion and how these variables affect drug reflux, needle contamination, vitreous incarceration, pain, scleral damage, and drug delivery have all been reported.
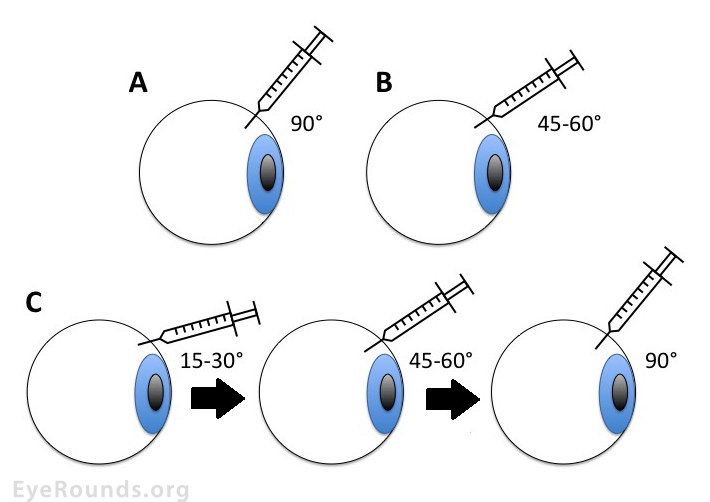
Several comparative studies of human eyes undergoing IVI report that vitreous reflux (measured by the size of post-IVI subconjunctival bleb) is significantly lower in tunneled scleral injection when compared to straight injection techniques [36-39]. There appears to be no difference in patient discomfort between tunneled and straight techniques [36 ,38], and there was no difference in IOP spike at 5 minutes after injection [38]. A more recent prospective study, by Özkaya et al., compared the effects of straight, oblique, and double-plane tunnel scleral IVI, on short-term IOP changes, vitreous reflux, and other complications [40]. They found that double-plane tunneled IVI prevents VR from the injection site and has no more complications than alternative techniques (Figure 2).
Larger 26- and 27-gauge needles are reported to cause greater vitreous reflux in comparison to 29- and 30-gauge needles. However, the width of the needle significantly affected the degree of reflux only when using the non-beveled incision [36]. Another study of IVI on porcine eyes found that 30-gauge needles showed less drug reflux than 32-gauge or 27-gauge. Thirty-two-gauge needles demonstrated less incarceration of vitreous at the tract site, but all needle tracts showed some internal vitreous incarceration (Figure 3). Deep IVI showed less reflux than superficial, but vitreous incarceration did not differ. Speed of injection did not modify the reflux or the vitreous incarceration [41]. Patients injected with the 26- or 27-gauge needle experienced more pain matched to the 29- and 30-gauge needles [36].
A study analyzing the effects of various needles on porcine eyes found less ocular damage and smaller scleral holes with increased needle gauge for both perpendicular (or straight) and tunneled injection. Statistical analysis of data showed higher probability of scleral opening reduction when a tunneled incision was used [39]. The same study found that residual drug volume was lower in small syringes of 0.3 mL connected to long 12.7-mm needles, implying that the popular use of 1-mL syringes may not be most appropriate. Needle size appears not to be a risk factor for contamination during IVT injection [42].
The mechanism of post-IVI elevation in IOP is likely due to increased volume of intraocular contents [43]. The spike is usually transient. Gismondi et al. found a significant difference in pre-IVI IOP and post-IVI IOP after five seconds to 30 minutes but not after one hour or one day [44]. Several other studies report similar findings, with most patients measuring increased IOP compared to baseline at 30 minutes post-IVI and then normal IOP by one week of follow-up [45]. Interestingly, there appears to be no difference in IOP for the type of medication injected, which often vary in volume [46]. There may be a relationship between axial length and very transient post-IVI spike in IOP, but data are conflicting. Gismondi et al. found a significant relationship between shorter axial length and post-ranibizumab IOP after five seconds but not one hour [44]. A more recent study using bevacizumab showed shorter eyes had a higher IOP at one and 15 minutes post-IVI [47]. Goktas et al., however, found no relationship between axial length or anterior chamber depth and post-ranibizumab IOP [48].
Occasionally, post-IVI IOP remains elevated. The exact mechanism of sustained ocular hypertension is controversial. Though sustained elevation in IOP is reported to occur in patients with no prior history of glaucoma, glaucoma suspect, or ocular hypertension (OHT), some studies suggest that patients with glaucoma may experience sustained IOP elevation at a greater rate than those without an existing diagnosis of glaucoma or OHT [45].
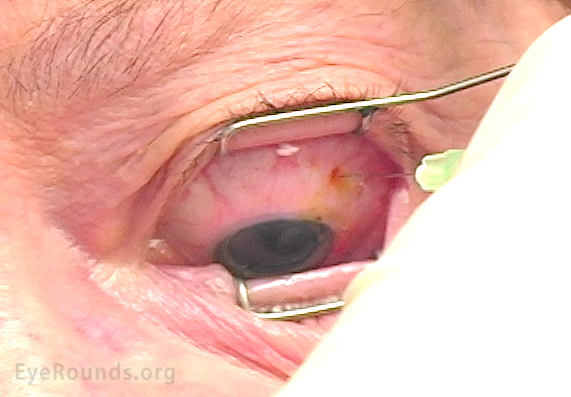
As with any other cause (or unknown cause) of elevated IOP, concern surrounds potential damage to the optic nerve resulting from decreased perfusion. Post-IVI temporary occlusion of central retinal artery, which quickly resolved with anterior paracentesis, has been reported [53]. In a 2011 survey of United States retinal specialists, nearly three-fourths of survey respondents (72%) routinely assess post-IVI optic nerve perfusion in some way. Of these, 32% perform a gross visual acuity examination by finger counting (Figure 4) or hand motion assessment, 21% visualize the optic nerve by indirect ophthalmoscopy, 15% measure the IOP, and 31% use a combination of these techniques [13].
The use of topical antibiotics is standard after ocular surgery. This principle was carried over to IVI, and logically so. In a 2011 survey of AAO retinal specialists, 81% (608/753) of respondents reported using post-IVI prophylactic antibiotics [13]. Topical antibiotics do reduce conjunctival bacterial growth [30], but no randomized controlled trials show a reduction in EO with post-IVI antibiotic use.
A low rate of EO can be achieved without topical antibiotics. Bhavsar et al. reported EO rates based on protocol requiring topical PI, sterile lid speculum, and topical anesthetic across four DRCR.net randomized trials. No topical antibiotics, sterile gloves, or sterile drapes were used. Of 8,027 IVIs, seven cases of EO occurred, and six of these seven received antibiotics [4].
A retrospective case control study of 117,171 IVI with or without antibiotics, reported that antibiotics do not seem to reduce EO but are actually associated with a trend toward higher incidence of EO, though the increased risk (odds ratio 1.54) was not statistically significant [54].
In a retrospective review of 15,895 IVIs of ranibizumab, bevacizumab, triamcinolone acetonide, or pegaptanib sodium where 9 eyes of 9 patients developed suspected EO (only 3 were culture positive), the incidence per injection was 0.06% for patients who were given 5 days of post-IVI antibiotics, 0.08% for those who received antibiotics immediately after IVI, and 0.04% for those receiving no antibiotics. However, statistical significance was not demonstrated [8].
Use of topical antibiotics may lead to increase in resistant organisms. Dave et al. demonstrated that eyes treated with post-IVI topical fluoroquinolones develop multi-drug resistant conjunctival flora (90% S. epidermidis) compared to controls (69% S. epidermidis) after just four IVIs (p < 0.02) [55]. Similarly, Milder et al. found that treated eyes had 87.5 % resistance to fluoroquinolones compared to 25% in controls (P = 0.04)[56]. In a non-randomized, prospective cohort study by Vin et al., the group receiving three days of post-IVI topical moxifloxacin had a higher culture-positive rate at one, two, and three months compared to the control group. MIC levels increased by 20% in the intervention group compared to a 5% decrease in the control group, and resistant isolates and MIC90 were approximately four times higher in the intervention group [57].
In a prospective, controlled longitudinal study of 24 patients (48 eyes), Dave et al. reported S. epidermidis and S. aureus comprise 54.5% and 18.2% of cultured isolates, respectively, prior to azithromycin exposure, and 90.9% (P<0.01) and 4.5% (P<0.01), respectively, after azithromycin exposure. In another group, 45.7% and 6.5% of isolates are S. epidermidis and S. aureus, respectively, at baseline, then 63.4% (P<0.03) and 13% (P = 0.24) after fluoroquinolone exposure [55].
The good news? Ocular surface preparation for IVI using PI 5% alone in the absence of post-injection topical antibiotics does not appear to promote bacterial resistance or a discernible change in conjunctival flora [58].
Many patients with bilateral disease, like choroidal neovascularization from age-related macular degeneration and diabetic macular edema, require IVI of both eyes. Same-day IVI is more convenient and cost-effective for patients, and many patients prefer bilateral injections [59]. Ophthalmologists must carefully weigh the risks and benefits of bilateral IVI. The most dreaded result of bilateral, same-day IVI is described in a 2013 case report of two patients who developed acute, bilateral EO following bilateral IVI [60].
A recent editorial by Chao et al. summarizes previous studies reporting bilateral, same-day IVI and associated EO incidence: From the University of Iowa, 102 patients, 452 injections, no EO [59]; From Bascom Palmer Eye Institute, 127 patients, 1,322 injections, no EO [61]; From a cohort in Korea, 135 patients, 574 injections, no EO [62]; From New York City cohort, 367 patients, 1,552 injections, one case of unilateral, culture-negative EO (0.033%) and two cases of culture-proven, unilateral EO (0.065%) [63]. They also give data from retina clinics at the Miami Veterans Affairs Hospital, where bilateral, same-day IVI are often performed, and performed by residents and fellows. From October 2007 to May 2014, 660 same-day, bilateral and 3,570 unilateral IVI were delivered. Patients undergoing same-day injections had each eye performed without reuse of any instruments or medications, meaning each eye was treated as though it belonged to a separate patient. There were zero cases of EO, and 438 of the 660 bilateral injections were delivered without topical antibiotics, as they discontinued use of topical antibiotics in August 2011. Masks, sterile gloves, and drapes were not used. They concluded, based on the data presented and data reviewed, that bilateral, same-day injections are safe [64].
The protocol described below is used at the Iowa City Veterans Affairs Medical Center (VAMC). There is minor variation in protocol at University of Iowa Hospitals and Clinics (UIHC), based primarily on specialist preference.
Video 1: UIHC technique for intravitreal injection. Use this link for alternative method of accessing video: https://vimeo.com/116066821.
Myers (Provencher) LA, Almeida D, Abràmoff MD. Intravitreal Injection Technique: A Primer for Ophthalmology Residents and Fellows. EyeRounds.org. January 6, 2015; Available from: http://www.EyeRounds.org/tutorials/intravitreal-injection/.