Headache and diplopia
A 38-year-old woman developed a headache across the forehead and behind the left eye that began five days prior to presentation. The headache worsened over the course of two days with onset of vomiting on day three. On day four, she developed left-sided facial swelling and an inability to open the left eye. Concomitantly on day four, she developed binocular diplopia, worse with attempted up-gaze that persisted until presentation. Upon initial evaluation, she rated her headache as a 7/10 and noted blurry vision and photophobia of the left eye. She denied neck stiffness, fever, or malaise.
Per HPI, otherwise negative
 |
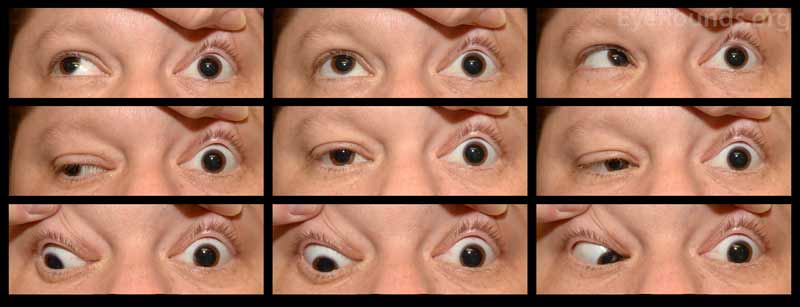 |
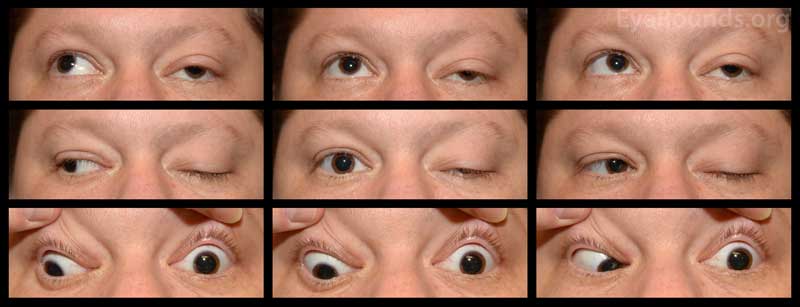
Figure 1: Nine gaze photos show left exotropia (10 PD) with a left hypotropia (25 PD) in primary gaze. Motility exam demonstrates a -3.5 gaze restriction in all cardinal gazes OS.
Figure 2: External photo was notable for left-sided ptosis accompanied by proptosis
 |
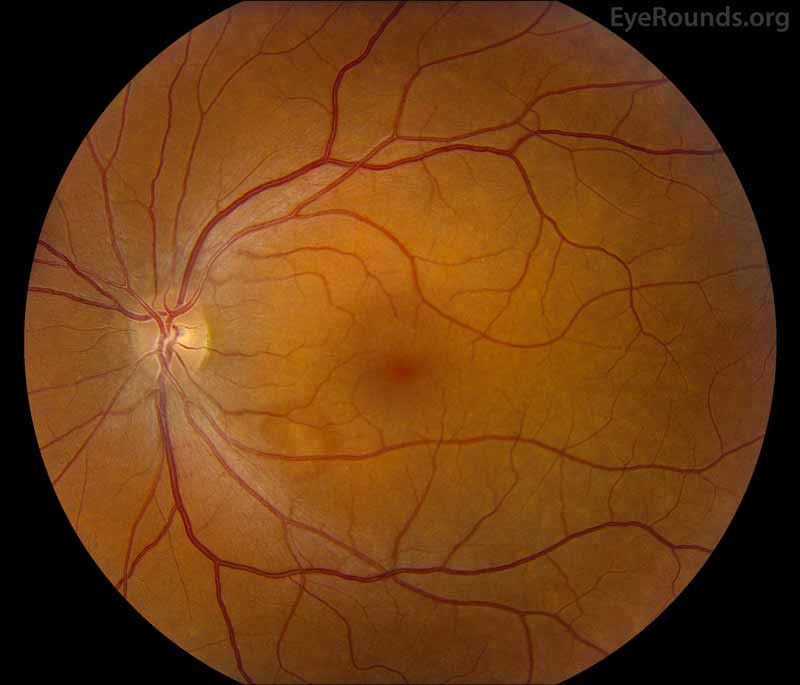 |
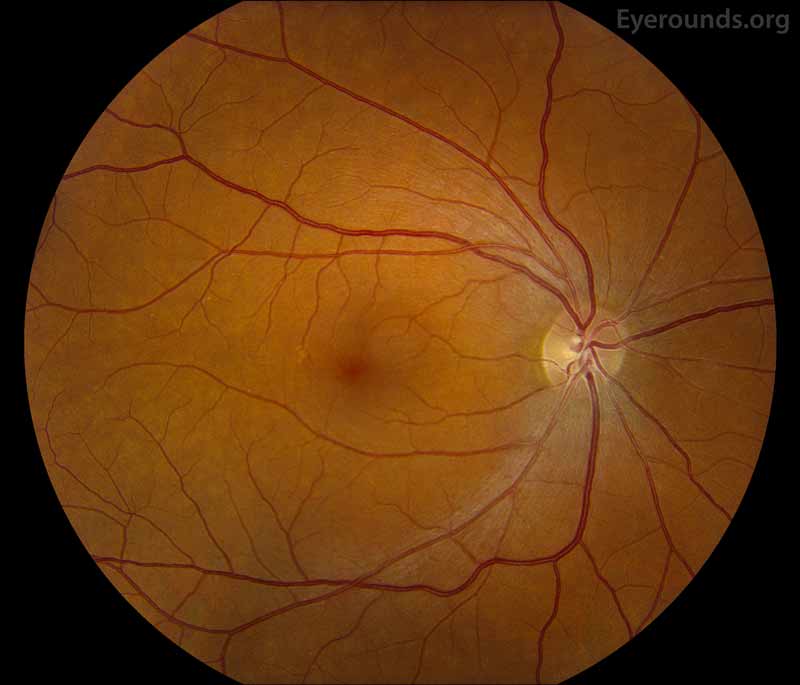
Figure 3: Fundus photos shows mild torsion of left eye (right image), otherwise normal in both eyes.
 |
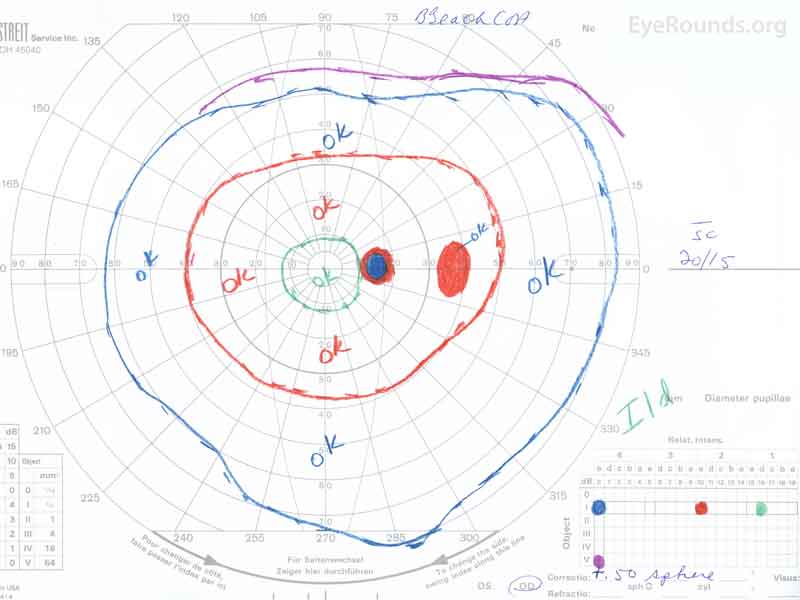 |
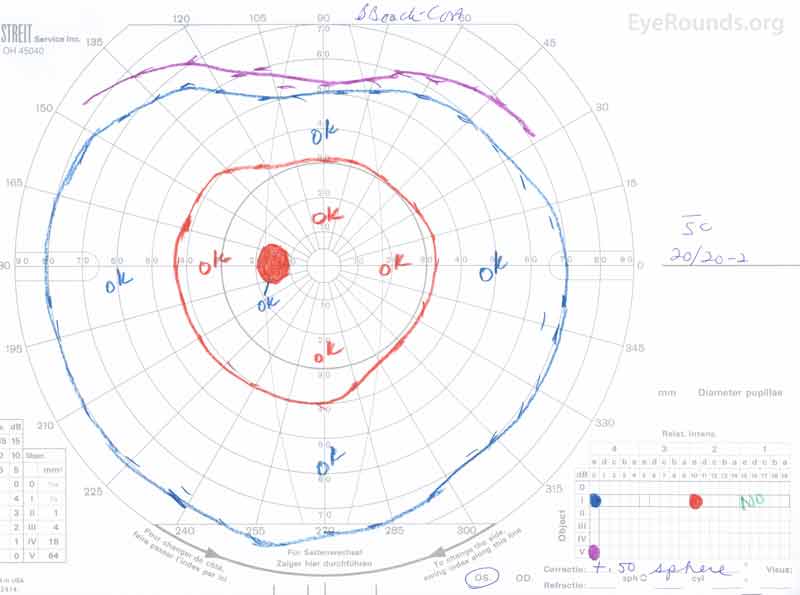
Figure 4: Goldmann kinetic perimetry of both eyes. OD: Full with a relative scotoma to the I2e isopter temporal to the blindspot. OS: Loss of the I1e isopter, otherwise full.
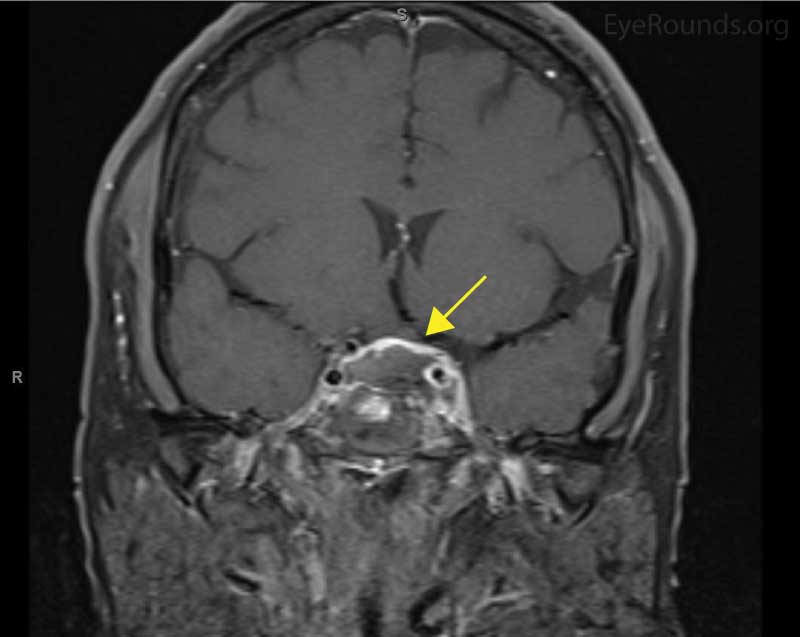
Coronal and sagittal T1 post-contrast MRI revealed a suprasellar lesion containing areas of suspected hemorrhage and necrosis. There was adjacent spread into the left cavernous sinus. A characteristic pituitary ring sign is suspicious for pituitary apoplexy (Figure 5).
 |
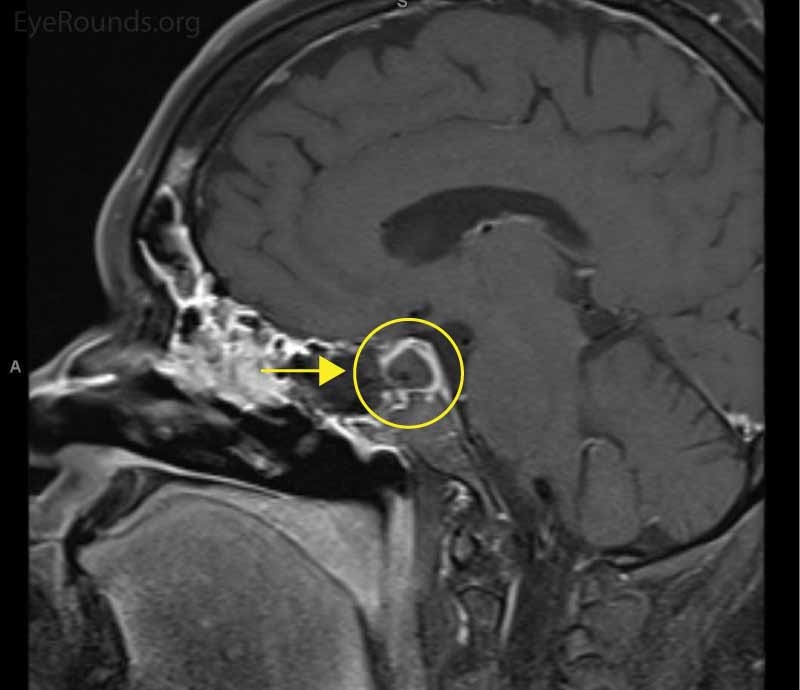 |
Figure 5: Magnetic resonance imaging of the brain. Left: Coronal image of a suprasellar lesion (arrow) with adjacent spread into the left cavernous sinus. Areas of suspected hemorrhage and necrosis are seen. Right: Sagittal view showing the characteristic pituitary ring sign (arrow), which is suspicious for pituitary apoplexy.
The patient underwent trans-sphenoidal decompression with trans-sphenoidal access provided by otorhinolaryngology and subsequent decompression performed by neurosurgery. Specimens were taken from the sellar mass due to concern for an infiltrative process, and the pathology was negative. Soon after decompression, her ptosis improved and CN function returned over the following weeks. No anterior visual pathway compression developed. Nearly full ocular motility was regained, with only a small amount of residual diplopia remaining in left gaze. Diabetes insipidus, a complication of pituitary dysfunction, developed during her convalescence and was treated with vasopressin.
Left-sided cavernous sinus syndrome affecting CN III, IV, V1, V2, and VI secondary to pituitary apoplexy
Cavernous sinus syndrome represents a constellation of signs and symptoms resulting from the impaired function of the neurologic structures running through this vascular space, namely CN III, IV, V1, V2, and VI, as well as the sympathetic nerves that innervate the iris dilator. The clinical presentation is marked by ptosis, ophthalmoplegia, and trigeminal dermatomal and corneal sensory loss. Depending upon the etiology, other external signs (e.g., proptosis, chemosis, conjunctival injection, etc.) and symptoms (e.g., headaches, pulsatile tinnitus, fever, malaise, weight loss, etc.) can be present. Similarly, an ipsilateral Horner's syndrome with VI nerve palsy would also localize to the cavernous sinus.
The etiologies responsible for cavernous sinus syndrome can be divided into three main categories: mass lesions, vascular etiologies, and granulomatous disease. The first category includes mass lesions compressing the cavernous sinus, such as hematomas, tumors (e.g., pituitary adenoma, meningioma, hemangioma, and metastases), and infections (e.g., bacterial abscesses or fungal masses such as mucormycosis or aspergillosis) [1-3]. The second category includes vascular etiologies such as carotid-cavernous fistulas, carotid artery aneurysms, or cavernous sinus thrombosis [1-3]. The third category is comprised of miscellaneous granulomatous diseases such as sarcoidosis, granulomatosis with polyangiitis, or Tolosa-Hunt syndrome [1-3].
Among vascular lesions, aneurysms typically cause indolent painful ophthalmoplegia, while fistulas and thrombosis may present acutely (e.g., direct carotid-cavernous fistula often related to trauma or aneurysm rupture) or with a slow, progressive course (e.g., indirect fistula usually involving dural branches of the carotid artery) [1, 4]. While some sources identify thrombosis related to microbial spread as the most common etiology [2], others report trauma and neoplasm as the most common etiologies of cavernous sinus syndrome [4-6].
Among the mass lesions capable of creating a cavernous sinus syndrome is a pituitary adenoma with or without apoplexy [7]. One study identified cavernous sinus invasion by pituitary tumors as a significant risk factor for pituitary apoplexy [8]. Therefore, when managing a patient with cavernous sinus syndrome, it is imperative to rule out and/or monitor for the development of pituitary apoplexy as severe hypoglycemia, hypotension, CNS hemorrhage, cardiovascular collapse, and death can result [9].
The cavernous sinus is an intracranial dural venous sinus. When relative blockage of one or both of the cavernous sinuses occurs, venous congestion in the superior and inferior orbital veins leads to chemosis and proptosis. Since the cavernous sinus and its walls contain a component of the third, fourth, fifth (ophthalmic and maxillary branches), and sixth CN as well as the carotid artery and sympathetic plexus, a lesion that impinges or irritates the cavernous sinus can affect some or all of these structures. Unilateral cavernous sinus syndrome may progress to become bilateral as two intercavernous sinuses connect the right and left cavernous sinuses [2].
Regardless of etiology, the CN deficits resulting from impingement or irritation of the cavernous sinus include ptosis (CN III), anisocoria (CN III), ophthalmoplegia (CN III and VI are affected in nearly all cases with CN IV affected in about one third of cases) [5], corneal sensory loss (CN V1), and middle and upper facial sensory loss (CN V1, CN V2). In addition, chemosis and proptosis are expected due to venous congestion distal to the cavernous sinus. Pain is often present, but when absent has been identified in one study as an independent risk factor for the presence of a tumor [6, 7]. If a vascular fistula is present, the typical triad of signs includes pulsatile exophthalmos, chemosis, and an orbital bruit [1]. Less common signs of a vascular etiology include ischemia to the eye or retina, choroidal effusions, or rarely cerebral venous infarction [10].
Cavernous sinus syndrome must be distinguished from pathology in the orbital apex. CN II involvement suggests orbital apex pathology but can occasionally be affected indirectly in a cavernous sinus syndrome [10]. For example, an extension of a tumor that begins near the cavernous sinus and extends toward the orbital apex may cause CN II involvement. One study demonstrated that all cases of cavernous sinus syndrome involving CN II were due to tumor [6]. Involvement of CN V2 indicates cavernous sinus syndrome while absence of CN V2 involvement indicates pathology in the orbital apex or superior orbital fissure. Since some cases may be impossible to clinically differentiate between the cavernous sinus and superior orbital fissure, the term sphenocavernous syndrome is sometimes used [10].
MRI is ideal when a mass lesion is suspected; specifically, the absence of bruit or pulsatile exophthalmos raises suspicion for mass lesions [4, 11]. MRI is also superior for visualization of the orbital apex and orbito-calvarial junction in addition to detection of cavernous sinus thrombosis [12]. If carotid artery-cavernous fistula is suspected, ultrasound may be utilized as a more cost-effective diagnostic tool where a skilled operator is available [12]. Cerebral CT angiography is usually used to distinguish a high-flow from a low-flow fistula [10, 12]. In order to provide sufficient detail for successful treatment of a carotid artery-cavernous sinus fistula, catheter angiography is needed and can be used simultaneously with treatment [12].
Treatment for cavernous sinus syndrome is dependent on the etiology. Cases of infectious thrombosis warrant prompt broad-spectrum intravenous antibiotics, accompanied by identification of the causal organism and drainage of any existing abscesses [1]. If the cavernous sinus syndrome is caused by infections such as Mucor, treatment is difficult but emergent surgical debridement and intravenous amphotericin B may be life-saving [4, 12-14]. Vascular causes require anticoagulation with or without interventional vascular repair [1]. Idiopathic granulomatous disease (e.g., Tolosa-Hunt syndrome) is treated with prednisone, typically 60 mg daily for 2 weeks followed by a month-long taper with methotrexate or azathioprine being added at the physician's discretion [1].
Treatment of mass lesions varies widely. In the case of a macroadenoma with or without apoplexy, dopamine agonist therapy should be started, typically with cabergoline [11]. Transsphenoidal resection is useful when dopamine therapy is unsuccessful or pregnancy is desired [11]. If apoplexy occurs, patients without impaired consciousness or visual loss can be managed conservatively with high-dose glucocorticoids. Progressive neurologic defect, loss of consciousness, or vision loss necessitates urgent surgical decompression [15].
EPIDEMIOLOGY / ETIOLOGYEtiologies include:
|
SIGNS
|
SYMPTOMS
|
TREATMENT
|
Evans JA, Clark TJE, Bailey JA. Cavernous Sinus Syndrome Secondary to Pituitary Apoplexy. EyeRounds.org. posted June 26, 2018; Available from https://EyeRounds.org/cases/271-cavernous-sinus-syndrome.htm

Ophthalmic Atlas Images by EyeRounds.org, The University of Iowa are licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License.