Adenoid cystic carcinoma of the lacrimal gland
101-year-old female with a 3-year history of a left orbital mass
Adenoid cystic carcinoma of the lacrimal gland
101-year-old female with a 3-year history of a left orbital mass
Chief Complaint
"Decreased vision in the left eye."
History of Present Illness (HPI)
A 99-year-old patient presented to the oculoplastics clinic with double vision, left proptosis, and decreased vision on the left, which had progressed slowly over the last year.
Past medical history
- Hypertension
- Hyperlipidemia
- Cardiac stents
- Uterine cancer in 1970, status-post hysterectomy and radiation treatment
Ocular history
- Cataracts surgery both eyes (OU)
Medications
- Aspirin
- Iron supplements
- Verapamil
- Isosorbide mononitrate
Allergies
Family History
Social history
Ocular exam
Visual Acuity
- Right eye (OD): 20/60 with correction, 20/25 pinhole
- Left eye (OS): 20/50 with correction, no improvement with pinhole
External
OS: Proptosis with inferonasal displacement of the globe
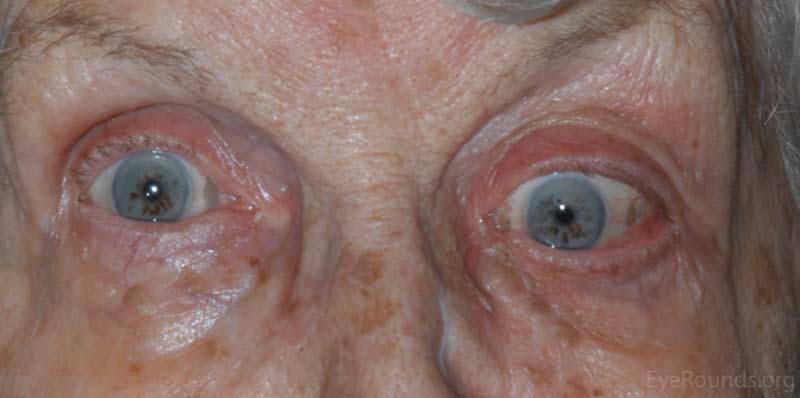
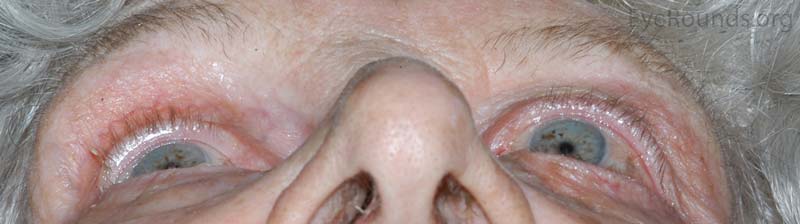
Figure 1: External photos demonstrates proptosis of the left eye with inferonasal displacement of the left globe.
Pupils
- OD: dark 3mm, light 2mm, no relative afferent pupillary defect (RAPD)
- OS: dark 3mm, light 2mm, no RAPD
Motility
- OD: Full
- OS: -1 limitation in infraduction, -2 limitation in adduction and supraduction, -3 limitation in abduction
Confrontational Visual Fields
Slit Lamp Exam
- Lids/Lashes: Normal OU
- Conjunctiva/Sclera: Clear and quiet OU
- Cornea: Clear OU
- Anterior Chamber: Deep and quiet OU
- Iris: Normal OU
- Lens: Posterior chamber intraocular lens (PCIOL) OU
Further workup
Imaging:
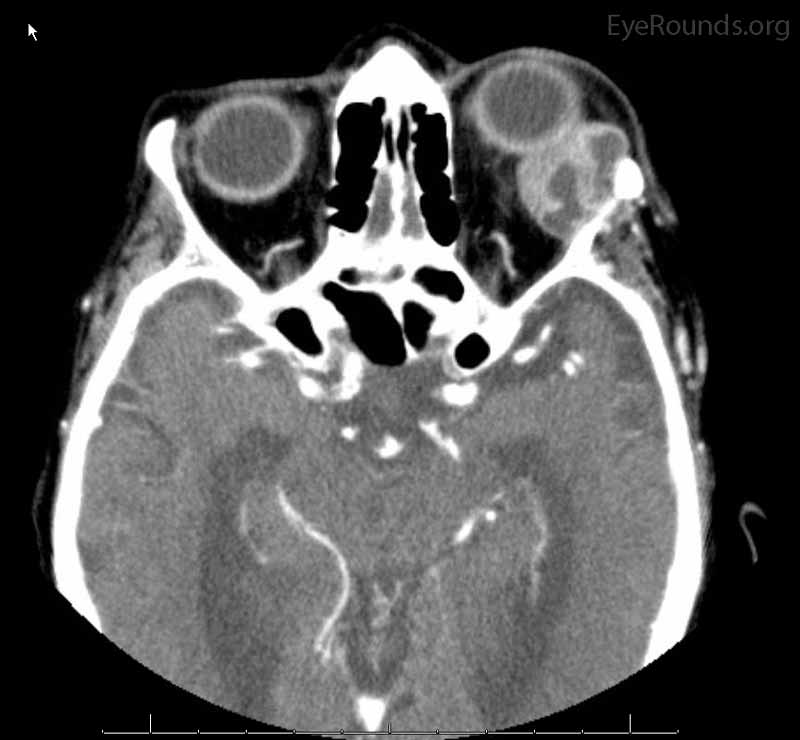
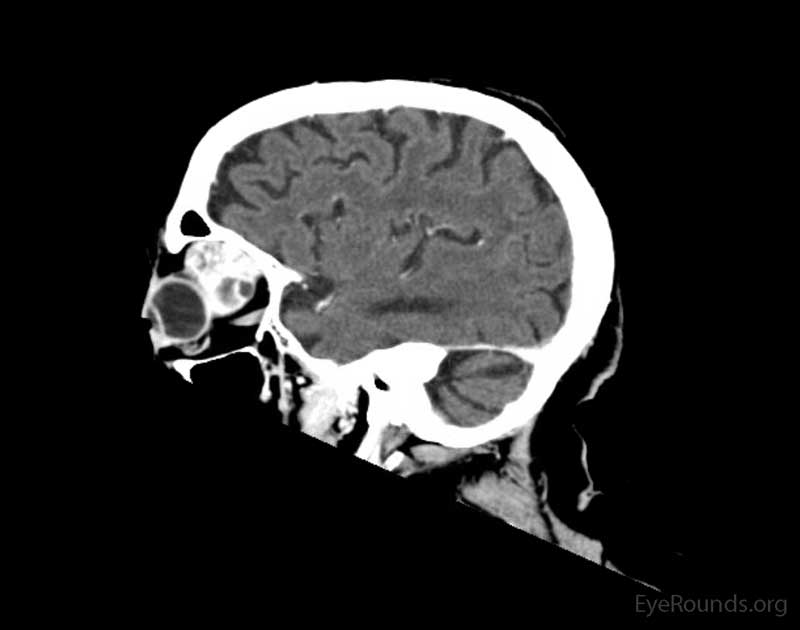
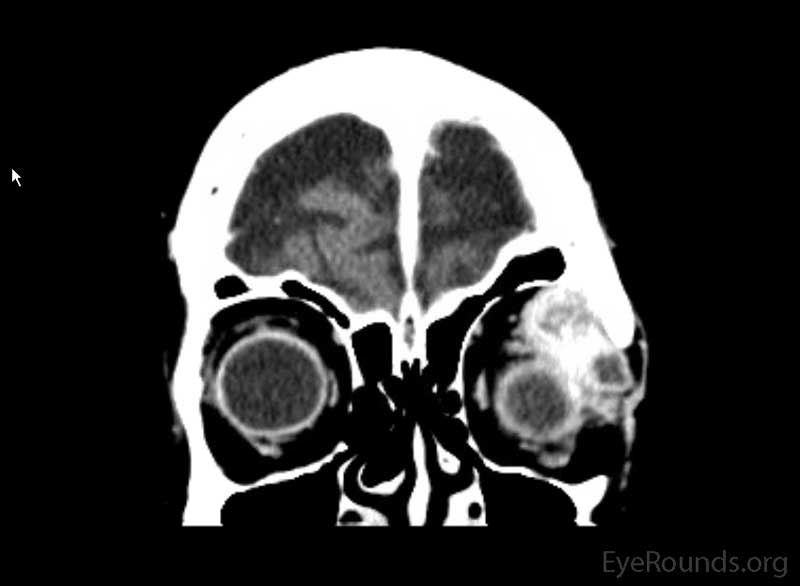
Figure 2: Coronal, frontal, and sagittal views from CT maxillofacial with contrast at the time of initial presentation. The CT demonstrates a left orbital mass displaying both solid enhancing and cystic components. The epicenter of the mass appears to be in the left lacrimal gland. There is left proptosis with medial displacement of the optic nerve and extraocular muscles. There is no definite sign of osteolysis of the orbital walls, however, there is bone remodeling with depression of the orbital floor. No evidence of intracranial extension of the left orbital mass.
Clinical course
The suspicion of an orbital process was confirmed after a CT maxillofacial scan showed a left orbital mass. Findings were suspicious for adenocarcinoma of the lacrimal gland. A left anterior orbitotomy was recommended, which the patient declined due to her age.
The patient was lost to regular follow up, however presented again two years after the initial presentation, at the age of 101-years-old. At this time she reported no vision in the left eye and growth of a now large mass which impaired her ability to socialize normally with other residents at her facility. She had no pain. Despite her age, she reported she re-presented because she desired to have the mass removed. She had no changed in her general health otherwise.
Ocular exam
Visual Acuity
- OD: 20/30-2 without correction, no improvement with pinhole
- OS: No light perception (NLP)
External
- OD: Normal
- OS: An extremely large 9cm x 8cm mass extending from the supraorbital area with inferonasal displacement of the globe. The lesion is irregular and the overlying skin is erythematous.
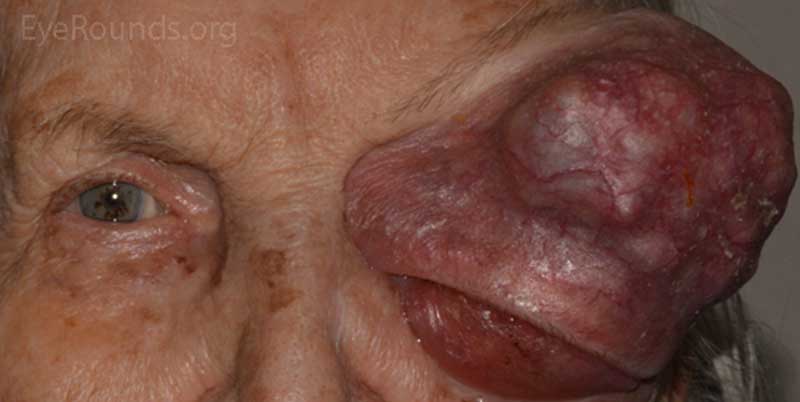
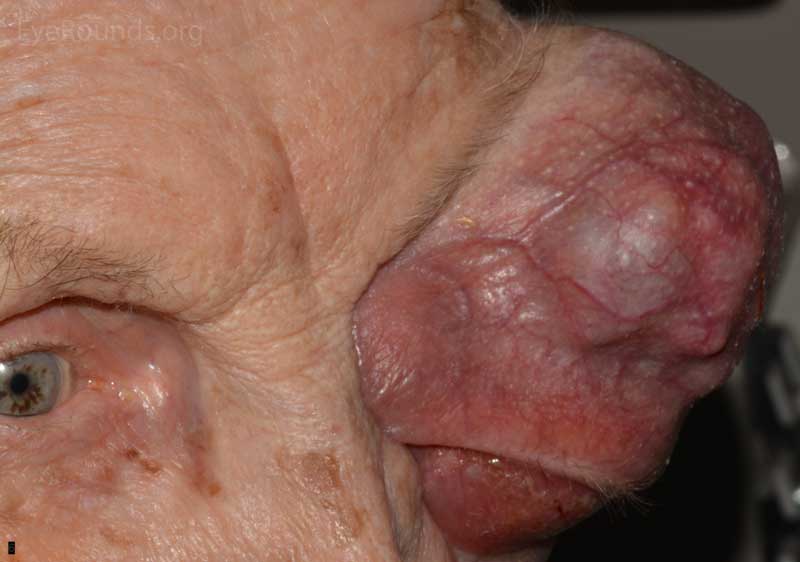
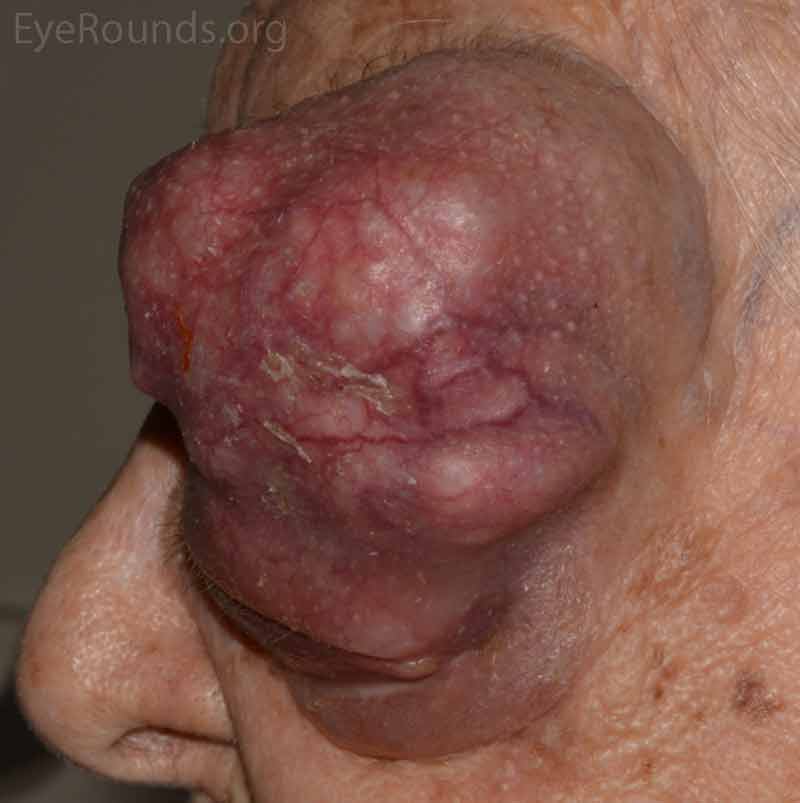
Figure 3: External photos demonstrates a large 9cm x 8cm mass extending from the supraorbital area with inferonasal displacement of the globe. The lesion is irregular and the overlying skin is erythematous. There is limited view of the left globe, which is displaced inferonasally.
Pupils
- OD: dark 3mm, light 2mm
- OS: dark 3mm, light 2mm, >3.0 log unit RAPD
Motility
- OD: Full
- OS: Complete ophthalmoplegia
Confrontational Visual Fields
- OD: Full
- OS: None (Vision is NLP)
Slit Lamp Exam
- Limited view due to extreme displacement of the globe inferonasally OS.
- Conjunctiva/Sclera: Clear and quiet OD
- Cornea: Clear OD
- Anterior Chamber: Deep and quiet OD
- Iris: Normal OD
- Lens: PCIOL OD
Further workup
Repeat imaging was performed.
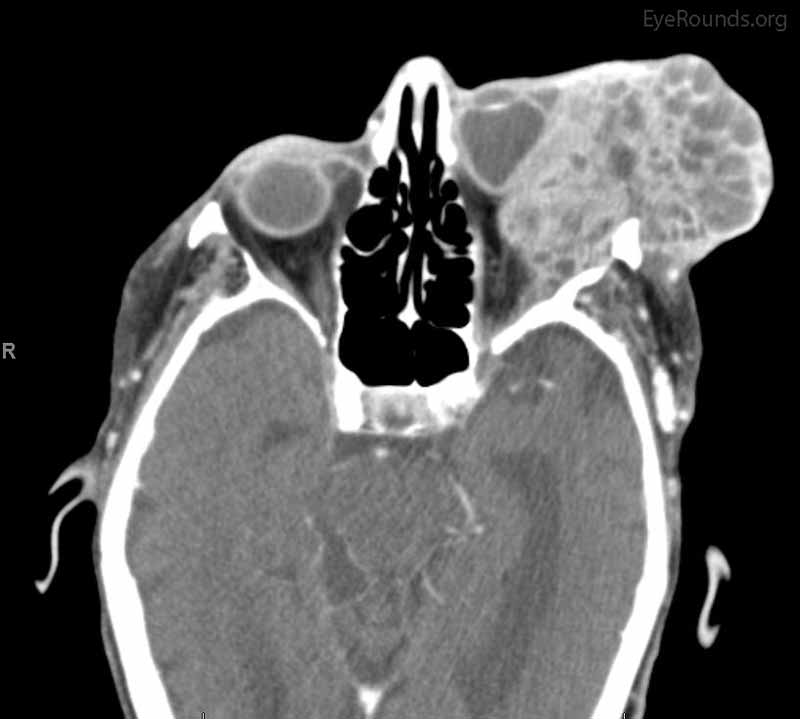
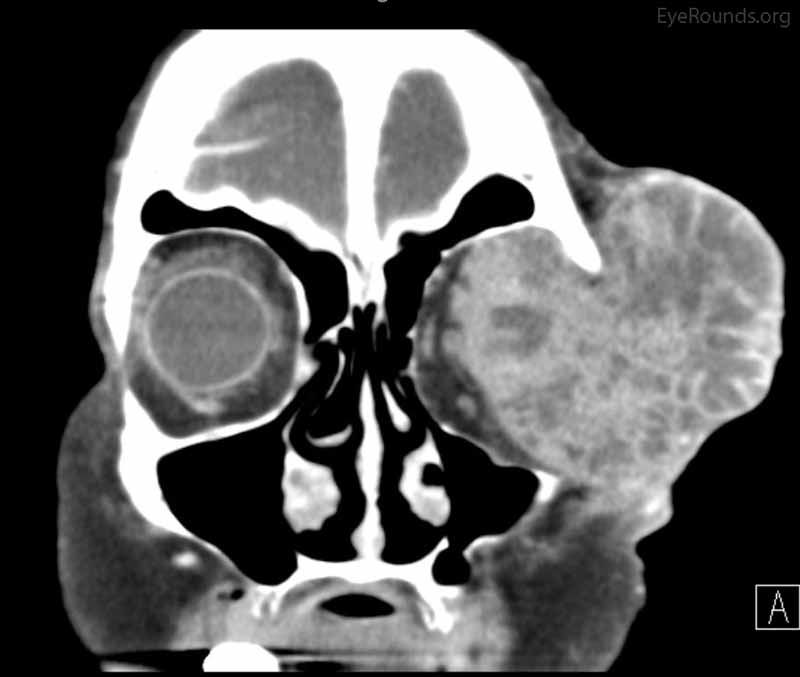
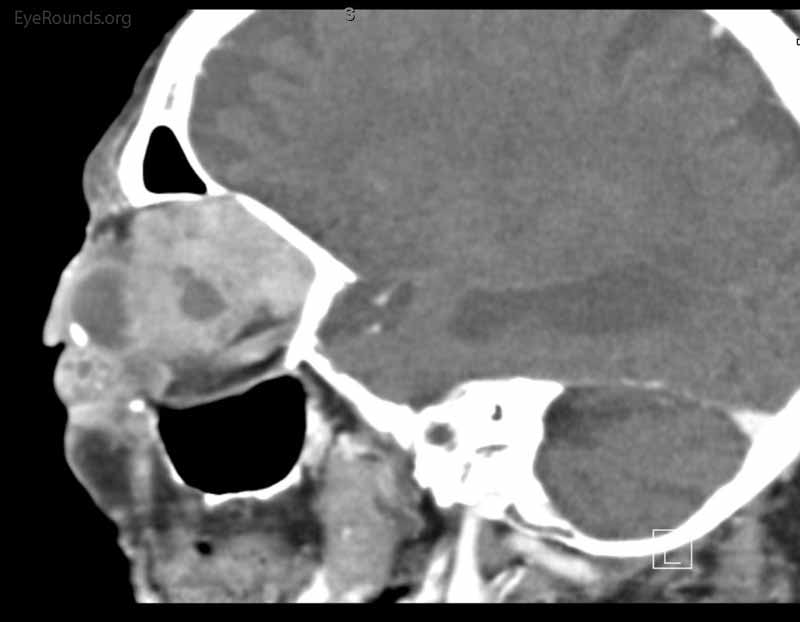
Figure 4: Coronal, frontal, and sagittal views from CT maxillofacial with contrast at the time of follow-up, The CT demonstrates massive interval growth of the left orbital mass displaying both solid enhancing and cystic components. The epicenter of the mass appears to be in the left lacrimal gland There is marked left proptosis with compression and deformation of the left globe as well as medial displacement of the optic nerve and extraocular muscles. There is no definite sign of osteolysis of the orbital walls, however, there is bone remodeling with depression of the orbital floor. No evidence of intracranial extension of the left orbital mass.
Clinical course
Given the patient's desire for removal, interval growth of the tumor, and NLP vision in the left eye, an exenteration of the left orbit was planned.
See the following surgical videos for an example of an exenteration
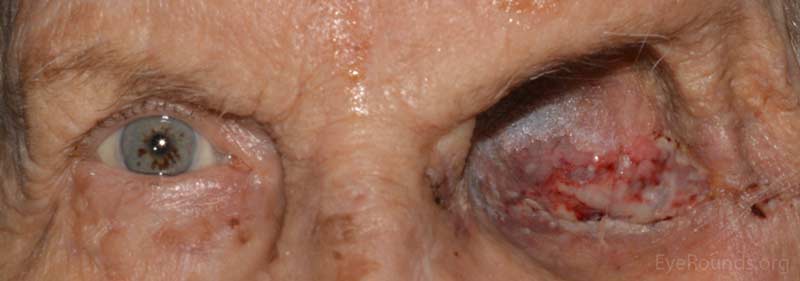
Figure 5: External photo demonstrates the left orbit at post operative month #1 after exenteration of the left orbital mass, which appeared to be granulating well.
Pathology
Left eye and orbital contents were sent to pathology following exenteration. Pathology demonstrated carcinoma infiltrating the ocular adnexa. The tumor displayed cribriform, cystoid, mucinous, and basaloid features. It was unclear whether this tumor was derived from the primary lacrimal gland or an accessory lacrimal gland. These findings were most consistent with adenoid cystic carcinoma of the lacrimal gland.
Diagnosis
Adenoid cystic carcinoma of the lacrimal gland
Discussion
Tumors of the lacrimal gland account for 10% of all space-occupying tumors of the orbit (1). Histologically, they are broadly characterized as epithelial and non-epithelial. The most common epithelial tumor is a benign pleomorphic adenoma, accounting for half of all epithelial tumors. The remaining half is malignant, with 20-30% identified as adenoid cystic carcinoma (ACC) (2). Overall, ACC accounts for 1.6% of all lacrimal gland tumors. Little is known regarding risk factors for development of ACC. Neither race nor geographic location contributes to risk and ACC is also equally prevalent among both genders (2). However, cigarette smoking and ionizing radiation have been reported as factors for corresponding similar carcinomas of the salivary glands (3).
ACC typically presents at a mean age of 50, although it is also found in children and the elderly (4). ACC can be slow growing, but with a more subacute worsening of symptoms (<1 year) than benign lesions of the lacrimal gland. Signs and symptoms include globe displacement, swelling/palpable mass, diplopia, and ptosis. The presentation may be subtle, and patients can often first present with only a complaint of pain. Up to 58-80% of patients with ACC complain of pain due to the tumor's tendency toward perineural invasion, particularly along the trigeminal nerve (5). One study reported up to 72% of tumor specimens showed evidence of perineural involvement (6).
A definitive diagnosis is obtained through tissue pathology, but imaging prior to tissue sampling is helpful in guiding the differential. On CT imaging, ACC typically appears solid with irregular margins and nodularity. Infiltration of adjacent tissue and bony destruction may be evident. MR imaging is most useful for assessing possible perineural invasion and extension into the cavernous sinus and brain. Moderate enhancement of the lesion and T1/T2 isointense to muscle are also features of ACC on MRI (7).
Treatment for ACC remains difficult with no general consensus on the most effective modalities. Typically, management involves tumor removal and post-operative radiotherapy. Surgical techniques depend on extension of tumor (bony involvement, perineural spread) and include local resection, enbloc removal, orbital exenteration, and radical exenteration. A retrospective analysis suggested that radiation therapy following surgical removal does improve overall survival rates, although this is contradicted in another study, thus further supporting the fact that standard treatment guidelines are difficult to propose (6, 8). Although resection can be successful at achieving local and regional control, it has not reduced the incidence of distant metastases (9). Newer studies have investigated intra-arterial chemotherapy, a previously available method, but not widely utilized. Intra-arterial chemotherapy, in addition to conventional methods (surgical resection, radiation), was found to reduce the carcinoma cause-specific death rate and 5-year recurrence rate when compared to conventional treatment alone (10, 11). Subsequent case reports have reported death remote from treatment due to relapse of disease in those treated with intra-arterial chemotherapy (12). Brachytherapy has also been implemented for incomplete resections, but data is limited for widespread use (13).
Prognosis remains grave for those diagnosed with ACC of the lacrimal gland. Overall 10-year survival rate was found to be between 20-40%, with variation in survival rates dependent on a few variables (4, 14). Difficulties in achieving complete surgical resection due to the complexity of the orbital anatomy lead to local recurrence, the most common site of recurrence. Mortality associated with ACC is typically due to intracranial spread or distant metastases (14). In addition to the brain, metastatic disease has been found in the lung, bone, liver, muscle, and spleen. Histopathologic tumor type has been shown to have prognostic implications. There are five identified tumor morphologies with microscopy, listed in order of most frequent to least: cribriform, basaloid, sclerosing, comedocarcinomatous, and tubular (15). A better prognosis is correlated to the cribriform variant, while basaloid findings in greater than 50% of pathologic specimens are associated with a significant decrease in disease-free survival (14, 16). Survival at 5 years for tumors with cribriform features was 71% compared with 21% for basaloid. Stage according to the AJCC Classification for Adenoid Cystic Carcinoma at diagnosis also influences survival rates. Those with stage ≥ T3 had a lower overall survival rate compared to those with <T3 staging. Additionally, those with stage ≥ T3 were found to have an increased incidence of local recurrence, distant metastases, and evidence of perineural spread, all factors that influence disease course (6). Age was found to be an independent predictor of overall survival and disease specific survival with the survival benefit conferred to younger patients (8). Another study has suggested that this is due to more favorable histopathologic features among this age group (17).
Differential diagnosis of lacrimal mass
- Neoplastic
- Vascular lesions
- Inflammatory lesion
EPIDEMIOLOGY
- Adenoid cystic carcinoma of the lacrimal gland accounts for 1.6% of orbital tumors
- Most common malignant epithelial lacrimal gland tumor
- No identified risk factors in the development of adenoid cystic carcinoma of the lacrimal gland
- Average age at diagnosis is 50; noted in children and elderly as well
|
SIGNS
- ± Palpable mass
- Decreased visual acuity
- Reduced ocular motility
- Facial asymmetry
- CT—typically solid lesion with irregular margins and nodularity. Infiltration of adjacent tissue and bony destruction may be evident.
- MRI—moderate enhancement of lesion. Best for assessing neural involvement.
|
SYMPTOMS
- Pain (cardinal symptom up to 80%)
- Facial asymmetry
- Ptosis
- Diplopia
|
TREATMENT
- Resection and post-operative radiation
- Intra-arterial chemotherapy may be used as an adjunct to surgical management/radiation therapy
- Brachytherapy has been used but is not common
|
References
- Shields CL, Shields JA, Eagle RC, Rathmell JP. Clinicopathologic review of 142 cases of lacrimal gland lesions. Ophthalmology. 1989;96(4):431-5.
- von Holstein SL, Coupland SE, Briscoe D, Le Tourneau C, Heegaard S. Epithelial tumours of the lacrimal gland: a clinical, histopathological, surgical and oncological survey. Acta Ophthalmol. 2013;91(3):195-206.
- Saku T, Hayashi Y, Takahara O, Matsuura H, Tokunaga M, Tokunaga M, et al. Salivary gland tumors among atomic bomb survivors, 1950-1987. Cancer. 1997;79(8):1465-75.
- Andreoli MT, Aakalu V, Setabutr P. Epidemiological trends in malignant lacrimal gland tumors. Otolaryngol Head Neck Surg. 2015;152(2):279-83.
- Bartley GB, Harris GJ. Adenoid cystic carcinoma of the lacrimal gland: is there a cure...yet? Ophthal Plast Reconstr Surg. 2002;18(5):315-8.
- Ahmad SM, Esmaeli B, Williams M, Nguyen J, Fay A, Woog J, et al. American Joint Committee on Cancer classification predicts outcome of patients with lacrimal gland adenoid cystic carcinoma. Ophthalmology. 2009;116(6):1210-5.
- Vaidhyanath R, Kirke R, Brown L, Sampath R. Lacrimal fossa lesions: pictorial review of CT and MRI features. Orbit. 2008;27(6):410-8.
- Mallen-St Clair J, Arshi A, Tajudeen B, Abemayor E, St John M. Epidemiology and treatment of lacrimal gland tumors: a population-based cohort analysis. JAMA Otolaryngol Head Neck Surg. 2014;140(12):1110-6.
- Esmaeli B, Golio D, Kies M, DeMonte F. Surgical management of locally advanced adenoid cystic carcinoma of the lacrimal gland. Ophthal Plast Reconstr Surg. 2006;22(5):366-70.
- Tse DT. Clinical and microdissection genotyping analyses of the effect of intra-arterial cytoreductive chemotherapy in the treatment of lacrimal gland adenoid cystic carcinoma. Trans Am Ophthalmol Soc. 2005;103:337-67.
- Tse DT, Benedetto P, Dubovy S, Schiffman JC, Feuer WJ. Clinical analysis of the effect of intraarterial cytoreductive chemotherapy in the treatment of lacrimal gland adenoid cystic carcinoma. Am J Ophthalmol. 2006;141(1):44-53.
- Fellman M, Carter K, Call CB, Esmaeli B. Disease recurrence after intraarterial chemotherapy in 2 patients with adenoid cystic carcinoma of lacrimal gland. Can J Ophthalmol. 2013;48(2):e17-8.
- Shields JA, Shields CL, Freire JE, Brady LW, Komarnicky L. Plaque radiotherapy for selected orbital malignancies: preliminary observations: the 2002 Montgomery Lecture, part 2. Ophthal Plast Reconstr Surg. 2003;19(2):91-5.
- Wright JE, Rose GE, Garner A. Primary malignant neoplasms of the lacrimal gland. Br J Ophthalmol. 1992;76(7):401-7.
- Tovilla-Canales JLB, S.; Olvera, O.; Martin, F.B. Diagnosis and Treatment of Lacrimal Gland Neoplasias: A better understanding of the nature of these tumors and advanced diagnostic technologies are improving management. Review of Ophthalmology. 2013.
- Gamel JW, Font RL. Adenoid cystic carcinoma of the lacrimal gland: the clinical significance of a basaloid histologic pattern. Hum Pathol. 1982;13(3):219-25.
- Tellado MV, McLean IW, Specht CS, Varga J. Adenoid cystic carcinomas of the lacrimal gland in childhood and adolescence. Ophthalmology. 1997;104(10):1622-5.
Suggested citation format:
Anderson LE, Beebe J, Allen RC. Adenoid cystic carcinoma of the lacrimal gland. EyeRounds.org. June 3, 2016. Available from https://eyerounds.org/cases/235-Adenoid-cystic-carcinoma-lacrimal-gland.htm
last updated: 06/03/2016
Image Permissions:

Ophthalmic Atlas Images by EyeRounds.org, The University of Iowa are licensed under a Creative Commons Attribution-NonCommercial-NoDerivs 3.0 Unported License.
